Advertisements
Advertisements
प्रश्न
Why carboxylic acid does not give reactions of carbonyl group?
Account for the following :
Carboxylic acids do not give reactions of carbonyl group.
उत्तर १
In carboxylic acid presence of lone pairs of electrons on oxygen which are involves in resonance due to this the electrophilic character of carbon in carboxylic acid decreases. So due to such reason carboxylic acid does not show the characteristic reaction of the carbonyl group.
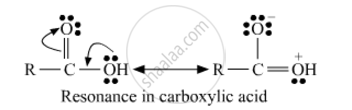
उत्तर २
In carboxylate anion, the carbon atom is joined to each oxygen atom by one and a half bond and the negative charge is distributed over both oxygen atoms. Thus, the carbonyl group in carboxylic acids is not a true carbonyl group as in aldehydes and ketones. Therefore, carboxylic acids do not give reactions of the carbonyl group.
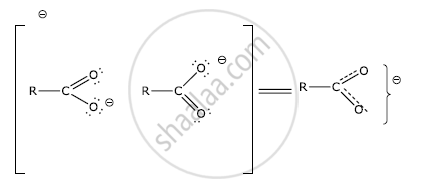
उत्तर ३
Carboxylic acids do not give reactions of carbonyl group because the lone pairs on oxygen attach to hydrogen in the –COOH group are involved in resonance which makes the carbon less electrophilic.
APPEARS IN
संबंधित प्रश्न
Draw the structure of the given derivative.
Acetaldehydedimethylacetal
Account for the following:
Aromatic carboxylic acids do not undergo Friedel-Crafts reaction.
\[\begin{array}{cc}
\ce{CH3 - C ≡ CH ->[40{%} H2SO4][1{%} HgSO4] A ->[Isomerisation] CH3 - C - CH3}\\
\phantom{........................................}||\\
\phantom{........................................}\ce{O}\\
\end{array}\]
Structure of ‘A’ and type of isomerism in the above reaction are respectively.
In HVZ reactions, the substitution takes place at:
Draw the structures of the given compound - 3-Bromo-4-phenylpentanoic acid
Draw the structure of the following compound.
3-Bromo-4-phenylpentanoic acid
Draw the structure of the following compound.
3-Bromo-4-phenylpentanoic acid
Draw the structure of the given compound.
3-Bromo-4-phenylpentanoic acid
Draw the structure of the following compound.
3-Bromo-4-phenylpentanoic acid
