Advertisements
Advertisements
प्रश्न
Why does the temperature remain constant during the boiling of water even though heat is supplied continuously ?
उत्तर
This is because the heat supplied is absorbed by the water particles and this heat increases the kinetic energy of all particles, which weakens the force of attraction between the particles and they start moving freely. As a result, water is converted into gas. This heat energy is called latent heat of vaporisation.
APPEARS IN
संबंधित प्रश्न
What is the physical state of water at 250°C.
What do you observe when :
some ice cubes are kept in a glass tumbler
Does the substance absorb or liberate any heat energy during the change of phase?
Draw a labelled diagram of the experimental set-up to study the latent heat of vaporisation of water.
Give two examples of the following:
Substances which do not change their states.
Explain the term condensation
Fill in the blank with the correct word Given below.
Cohesive forces are negligible in ____
Matter in any state is composed of small particles – molecules, atoms, or ions. Differentiate the terms above in Italics.
Give the reason for the following
On heating a sublimable solid, the molecules break free & escape from the surface of the solid directly into vapour.
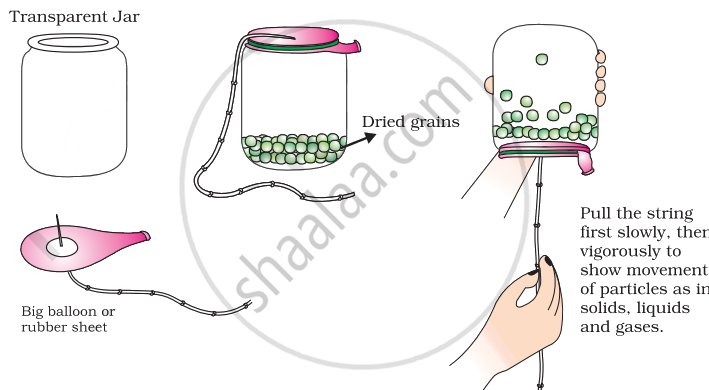
Prepare a model to demonstrate movement of particles in solids, liquids and gases.
For making this model you will need
- A transparent jar
- A big rubber balloon or piece of stretchable rubber sheet
- A string
- Few chickpeas or black gram or dry green peas.
How to make?
- Put the seeds in the jar.
- Sew the string to the centre of the rubber sheet and put some tape to keep it tied securely.
- Stretch and tie the rubber sheet on the mouth of the jar.
- Your model is ready. Now run your fingers up and down the string by first tugging at it slowly and then rapidly.