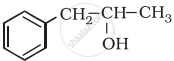Advertisements
Advertisements
प्रश्न
Why ethanol has the higher boiling point than ethane?
उत्तर
1) Alcohol has higher boiling points than their corresponding alkanes due to the presence of intermolecular hydrogen bonding which is absent in the alkanes.
2) Hydrogen bonding arises due to the presence of electronegative atom oxygen in –OH group of the alcohol.
3) Oxygen atom attracts electron density of O–H bond towards itself and hence it gets the partial negative charge, while H atom gets partial positive change.
`-delta + delta`
R-O-H
4) Hence in alcohol, R–OH molecules become polar
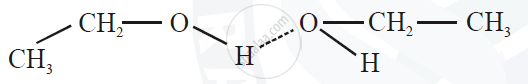
5) There arises strong intermolecular attraction between the oxygen atom of one molecule of alcohol and ‘H’ atom of another alcohol molecule, giving rise to strong hydrogen bonding which is not present in alkanes.
6) Hence higher thermal energy is required to separate or evaporate alcohol molecule. Therefore alcohols have higher boiling points than their corresponding alkanes.
APPEARS IN
संबंधित प्रश्न
Which of the followings is a trihydric alcohol ?
(a) n-propyl alcohol
(b) Glycerol
(c) Glycol
(d) Glycine
Classify the following as primary, secondary and tertiary alcohol:
\[\begin{array}{cc}
\ce{CH3}\phantom{.}\\
|\phantom{....}\\
\ce{CH3 - C - CH2OH}\\
|\phantom{....}\\
\ce{CH3}\phantom{.}
\end{array}\]
The alcohol used in thermometers is _______.
(A) methanol
(B) ethanol
(C) propanol
(D) butanol
Write a chemical equation for the action of neutral ferric chloride on phenol.
Write balanced chemical equation for the following :
Action of sodium metal on ethanol.
How many isomers of C5H11OH will be primary alcohols?
How many alcohol(s) with molecular formula \[\ce{C4H10O}\] is chiral in nature?
Benzyl alcohol is obtained from benzaldehyde by:
Among the following compounds, the strongest acid is:
Lower molecular mass alcohols are ______.
Which of the following compounds gives a secondary alcohol upon reaction with methylmagnesium bromide?
\[\ce{n-Butane ->[AlCl3][HCl] [X] ->[KMnO4] [Y], [Y]}\] is
Glycerol is not used in which of the following cares
Main constituent of dynamite is.
Write IUPAC names of the following compound:
\[\begin{array}{cc}
\phantom{..............}\ce{CH3}\\
\phantom{............}|\\
\ce{CH3 - CH - CH - C - CH3}\\
|\phantom{......}|\phantom{......}|\\
\phantom{...}\ce{CH3}\phantom{...}\ce{OH}\phantom{...}\ce{CH3}
\end{array}\]
Write IUPAC names of the following compounds:
\[\begin{array}{cc}
\phantom{...............}\ce{CH3}\\
\phantom{.............}|\\
\ce{CH3 - CH - CH - C - CH3}\\
|\phantom{......}|\phantom{......}|\\
\phantom{...}\ce{CH3 \phantom{..}OH \phantom{..}CH3}
\end{array}\]
Classify the following as primary, secondary and tertiary alcohol:
CH3 – CH2 – CH2 – OH
Classify the following as primary, secondary and tertiary alcohol: