Advertisements
Advertisements
प्रश्न
Why first ionisation enthalpy of Cr is lower than that of Zn?
उत्तर
The electronic configuration of chromium and zinc are respectively:
\[\ce{Cr (24) = [Ar] 3d^5 4s^2}\]
\[\ce{Zn (30) = [Ar] 3d^10 4s^2}\]
It is easy to remove electron from 4s1-orbital (unpaired) rather than from 4s2-orbital (paired).
APPEARS IN
संबंधित प्रश्न
Account for the following:
E° value for the Mn3+/Mn2+ couple is much more positive than that for Cr3+/Cr2+.
What may be the stable oxidation state of the transition element with the following d electron configurations in the ground state of their atoms?
3d3, 3d5, 3d8 and 3d4
An antifriction alloy made up of antimony with tin and copper, which is extensively used in machine bearings is called _______.
(A) Duralumin
(B) Babbitt metal
(C) Spiegeleisen
(D) Amalgam
Transition metals with highest melting point is ____________.
Generally transition elements form coloured salts due to the presence of unpaired electrons. Which of the following compounds will be coloured in solid-state?
Which of the following will not act as oxidising agents?
(i) \[\ce{CrO3}\]
(ii) \[\ce{MoO3}\]
(iii) \[\ce{WO3}\]
(iv) \[\ce{CrO^{2-}4}\]
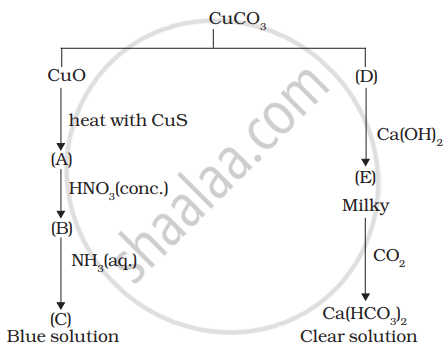
Identify A to E and also explain the reactions involved.
Assertion (A): Transition metals have high enthalpy of atomisation.
Reason (R): Greater number of unpaired electrons in transition metals results in weak metallic bonding.
Why are interstitial compounds well known for transition metals?
Compare the general characteristics of the first series of the transition metals with those of the second and third series metals in the respective vertical columns. Give special emphasis on the following point:
Atomic sizes
