Advertisements
Advertisements
प्रश्न
Write a balanced chemical equation for the following:
A mixture of sodalime and sodium acetate is heated.
उत्तर
\[\ce{\underset{Soda}{CH3COONa} + \underset{acetate}{NaOH} ->[Cao][Heat] \underset{Methane}{CH4} + \underset{Ethanoate}{Na2CO3}}\]
APPEARS IN
संबंधित प्रश्न
A student puts a drop of reaction mixture of a saponification reaction first a blue litmus paper and then on a red litmus paper. He may observe that:
(a) There is no change in the blue litmus paper and the red litmus paper turns white.
(b) There is no change in the red litmus paper and the blue litmus paper turns red.
(c) There is no change in the blue litmus paper and the red litmus paper turns blue.
(d) No change in colour is observed in both the litmus papers
A student adds 2 mL of acetic acid to a test tube containing 2 mL of distilled water. He then shakes the test tube well and leaves it to settle for some time. After about 5 minutes he observes that in the test tube there is :
(A) a clear transparent colourless solution
(B) a clear transparent pink solution
(C) a precipitate settling at the bottom of the test tube
(D) a layer of water the layer of acetic acid
Complete the following chemical equations: CH3COOH+NaHCO3→
Draw the structures for the following compounds of Propanoic acid.
What type of compound is formed when a carboxylic acid reacts with an alcohol in the presence of conc. H2SO4?
A student is studying the properties of acetic acid in his school laboratory. List two physical and two chemical properties which he must observe and note in his record book.
Write the important uses of acetic acid.
Fill in the blank with appropriate word/words.
Denatured alcohol is a mixture of _____ and _______
Write the chemical equation for the following:
Saponification Reaction
Observe the diagram given below and answer the questions:
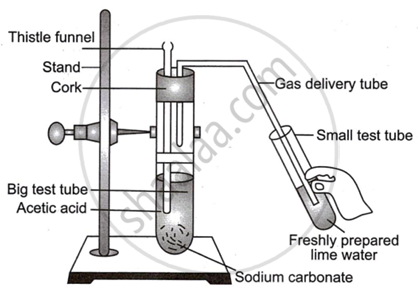
- Name the reactants in this reaction.
- Which gas comes out as effervescence in the bigger test tube?
- What is the colour change in the lime water?
- In the above experiment instead of sodium carbonate which chemical can be used to get same products?
- Write the use of acetic acid.
