Advertisements
Advertisements
प्रश्न
Write a reaction to distinguish acetaldehyde from acetone
उत्तर १
Tollens’ reagent test: Acetaldehyde being an aldehyde reduces Tollens’ reagent to shining silver mirror, whereas propanone being acetone does not.
\[\ce{\underset{\text{Acetaldehyde}}{CH3CHO} + \underset{\text{Tollens’ reagent}}{2[Ag(NH3)2]+} + 3OH- -> \underset{\text{Acetate ion}}{CH3COO-} + \underset{\text{Silver metal}}{2Ag ↓} + 4NH3 + 2H2O}\]
\[\ce{\underset{\text{Acetone}}{CH3COCH3} ->[Tollens 'reagent] No silver mirror}\]
उत्तर २
Fehling’s solution test: Acetaldehyde reduces Fehling’s solution to red precipitate of Cu2O whereas acetone being a ketone does not.
\[\ce{\underset{\text{Acetaldehyde}}{CH3CHO} + \underset{\text{From Fehling’s solution}}{2Cu^2+ + 5OH-} ->\underset{\text{Acetate ion}}{CH3COO-} + \underset{\text{Cuprous oxide (Red ppt.)}}{Cu2O ↓} + 3H2O}\]
APPEARS IN
संबंधित प्रश्न
The following compounds will give positive Fehling’s test.
Write another name of disproportionation reaction?
Identify B in the following reaction,
\[\ce{Acetaldoxime ->[Na][alcohol] A ->[NaNO2][HCl] B + H2O + N2\uparrow}\]
Which of following is not a property of red phosphorus?
Following reaction is an example of ____________.
\[\ce{Benzaldehyde ->[Nitration] {m}-Nitrobenzaldehyde}\]
The number of α-H atoms in butanal is ____________.
Which of the following carbonyl compounds does NOT undergo aldol condensation?
Identify the product Y in the following series of reactions.
\[\ce{4-Nitrotoluene ->[(CH3CO)2O/CrO3][273 - 278 K] X ->[H3O^+][\Delta] Y}\]
Reaction of acetone with one of the following reagents involves nucleophilic addition followed by elimination of water. The reagent is:
Assertion: 2, 2-dimethyl propanoic acid does not give HVZ reaction.
Reason: 2-2, dimethyl propanoic acid does not have α-hydrogen atom.
Which one of the following reaction is an example of disproportionation reaction.
Which one of the following undergoes reaction with 50% sodium hydroxide solution to give the corresponding alcohol and acid.
Carboxylic acids have higher boiling points than aldehydes, ketones and even alcohols of comparable molecular mass. It is due to their ____________.
What is the action of HCN on propanone?
What is the action of HCN on 2, 4-dichlorobenzaldehyde?
Identify A, B and C.
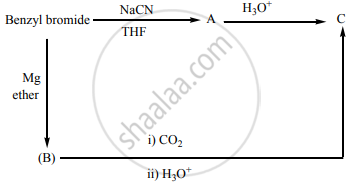
How propanal is converted into propane.
A substance C4H10O yields on oxidation a compound, C4H8O which gives an oxime and a positive iodoform test. The original substance on treatment with a conc. H2SO4 gives C4H8. The structure of the compound is ______.
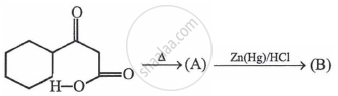
In the above reaction, product (B) is:
Which of the following is most reactive towards the addition reaction of hydrogen cyanide to form the corresponding cyanohydrin?
Which of the following is not correctly matched?
A carbonyl compound A having molecular formula C5H10O forms crystalline precipitate with sodium bisulphate and gives positive iodoform test. A does not reduce Fehling solution. Identify A.
Write reactions when phenol reacts with Dilute HNO3
Write the structure of the major product of the aldol condensation of benzaldehyde with acetone.
How will you prepare Acetic anhydride from acetic acid ?
Write the structure of the major product of the aldol condensation of benzaldehyde with acetone.
