Advertisements
Advertisements
प्रश्न
Write an equation for the reaction of hydrochloric acid on caustic soda solution.
उत्तर
\[\ce{NaOH + HCl -> NaCl + H2O}\]
APPEARS IN
संबंधित प्रश्न
Potassium sulfite on reacting with hydrochloric acid releases _______ gas. (Cl2, SO2, H2S)
The acid on mixing with silver nitrate solution produces a white precipitate which is soluble in excess ammonium hydroxide
State your observation When moist starch iodide paper is introduced into chlorine gas.
Write an equation for the reaction of hydrochloric acid on copper oxide.
Name the following:
A chemical in which gold can be dissolved.
Name the following:
A metallic oxide which reacts with hydrochloric acid to give a coloured solution.
Write balanced equcation for the following reaction:
Manganese (IV) oxide and concentrated hydrochloric acid.
State which of the two - a solution of HCl in water or in toluene is an electrolyte. Explain.
State which of the two - a solution of HCl in water or in toluene is an electrolyte. Explain.
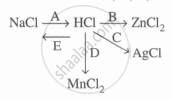
Study the flow chart and give balanced equations with conditions for the conversions A, B, C, D, and E.