Advertisements
Advertisements
प्रश्न
Write down the electronic configuration of the following
- 2713X,
- 3517Y.
Write down the number of electrons in X and neutrons in Y and the formula of the compound formed by X and Y.[XY3]
उत्तर
Electronic configuration of:
(1) 2713X number of electrons = 13
Distribution of elecrtrons in different ordits
13 = [2, 8, 3]
K L M
Number of neutrons in Y
= A − Z = 35 − 17 = 18
(2) 3517Y number of electrons = 17
Distribution of electrons in different orbits
17 = [2, 8, 7]
K L M
Formula of compound formed is
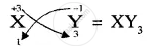
APPEARS IN
संबंधित प्रश्न
Fill in the blanks of the following statements :
An atom has atomic mass number 23 and atomic number 11. The atom has_________ electrons.
Cathode rays are a beam of fast moving _________
The maximum number of electrons that can be accommodated in L shell are_________
The maximum number of electrons that can go into the M shell is _________
How does an electron differ from a neutron?
Write down the electronic configuration of the following:
(a) `""_13^27"Y"`
(b) `""_17^35"Y"`
In the formation of (i) oxygen molecule (ii) carbon tetrachloride molecule, state the following:
- Electronic configuration of nearest inert gas attained.
- How many electrons are shared/transferred in bond formation
- Which type of bonds these compounds form?
- Draw their orbital diagrams.
The number of electrons in an element X is 15 and the number of neutrons is 16. Which of the following is the correct representation of the element?
Which of the following statement is always correct?
Atoms of all metals will have ______ electrons in their outermost orbit.
All non–metals will have ______ electrons in the outermost orbit of their atoms.
The relative charge of an electron is – 1.
In a television tube, Cathode rays are deflected by magnetic fields.
