Advertisements
Advertisements
प्रश्न
Write observation:
A small piece of zinc is added to dilute hydrochloric acid.
उत्तर
A gas (hydrogen) is evolved, which burns with a pop sound when exposed to air.
\[\ce{Zn + 2HCl -> ZnCl2 + H2 ^}\]
APPEARS IN
संबंधित प्रश्न
State your observation when dilute hydrochloric acid is added to a lead nitrate solution and the mixture is heated.
For the preparation of hydrochloric acid in the laboratory:
What arrangement is done to dissolve hydrogen chloride gas in water?
Give a chemical test to distinguish between the given pairs of chemicals:
Sodium chloride solution and Sodium nitrate solution
Write an equation for the reaction of hydrochloric acid on zinc carbonate.
Write an equation for the reaction of hydrochloric acid on copper oxide.
What is the function of HCI in preparation of aqua-regia?
Write a fully balanced of the following case :
Magnesium metal is treated with dilute hydrochloric acid.
Match the following:
| Column A | Column B |
| 1. A substance that turns moist starch iodide paper blue. | A. Ammonium sulphate |
| 2. A compound which release a reddish brown gas on reaction with concentrated sulphuric acid and copper turnings. | B. Lead carbonate |
| 3. A solution of this compound gives dirty green precipitate with sodium hydroxide. | C. Chlorine |
| 4. A compound which on heating with sodium hydroxide produces a gas which forms dense white fumes with hydrogen chloride. | D. Copper nitrate |
| 5. A white solid which gives a yellow residue on heating | E. Ferrous sulphate |
State which of the two - a solution of HCl in water or in toluene is an electrolyte. Explain.
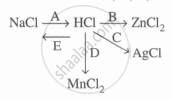
Study the flow chart and give balanced equations with conditions for the conversions A, B, C, D, and E.