Advertisements
Advertisements
प्रश्न
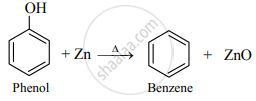
Write the balanced chemical reaction to get benzene from Phenol.
उत्तर
Phenol to benzene:
APPEARS IN
संबंधित प्रश्न
Name the following:
The hydrocarbon said to possess carcinogenic property.
Write the balanced chemical reaction to get benzene from Sodium benzoate.
Identify A, B, C in the following reaction sequence:
\[\ce{CH3 - CH = CH2 ->[Br2/CCl4][room temperature] A ->[Zn] B ->[dil. alkaline][KMnO4] C}\]
Identify giving reason whether the following compound is aromatic or not.
Read the following reaction and answer the questions given below:
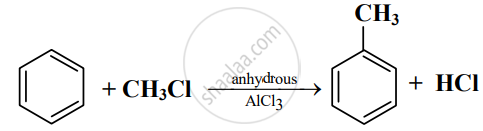
- Write the name of the reaction.
- Identify the electrophile in it.
- How is this electrophile generated?
Major product of the below mentioned reaction is, \[\ce{(CH3 )2 C = CH2 ->[ICI]}\]
Identify the compound (A) in the following reaction
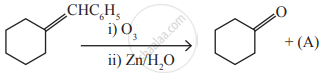
Which one of the following is non aromatic?
Identify the compound 'Z' in the following reaction
\[\ce{C2H6O ->[Al2O3][623 K] X ->[O3] Y ->[Zn/H2O] (Z)}\]
2 – butyne on chlorination gives ______.
Write the chemical equations for combustion of propane.
How does Huckel rule help to decide the aromatic character of a compound?
What happens when ethylene is passed through cold dilute alkaline potassium permanganate.

Identify A and B.
Which of the following is an aromatic compound?
A compound with molecular formula C4H4O has all the four carbon atom and the oxygen atom in the ring. It also has two carbon-carbon double bonds. The compound is ____________.
−Cl group is ____________.
According to Huckel rule, a cyclic π molecular orbital formed by overlap of p orbitals must contain ____________ p electrons.
Direct bromination of benzene with excess reagent results in the formation of ____________.
Sodium benzoate on decarboxylation gives ____________.
α-butylene when subjected to hydroboration oxidation reaction, yields ____________.
Conversion of hexane into benzene involves the reaction of ______.
Arenes on treatments with chlorine in the presence of ferric chloride as a catalyst undergo what type of reaction?
Identify the X and Y in the following reactions.
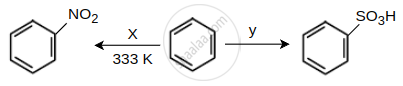
Read the following reaction and answer the questions given below.
\[\begin{array}{cc}
\phantom{..............................}\ce{CH3}\\
\phantom{............................}|\\
\ce{CH3 - C = CH2 + HBr ->[benzoyl][peroxide] CH3 - CH - CH2Br}\\
|\phantom{....................................}\\
\ce{CH_3}\phantom{.................................}
\end{array}\]
- Write the IUPAC name of the product.
- State the rule that governs the formation of this product.
