Advertisements
Advertisements
प्रश्न
Write the characteristics of ethanol.
उत्तर
- It is colourless liquid at room temperature and
- Its boiling point is 78 °C.
- It is soluble in water in all proportions.
- Its aqueous solution is neutral.
APPEARS IN
संबंधित प्रश्न
Write the name and structural formula of the compound obtained when ethanol is heated at 443 K with excess of conc. H2SO4. Also write chemical equation for the reaction stating the role of conc. H2SO4 in it.
The compound formed when ethene reacts with Hydrogen is ________. (CH4, C2H6, C3H8)
Give a balanced chemical equation for Preparation of ethane from Sodium propionate
What is the common name of methanal?
Complete the following equation:
`CH_3 CH_2 OH` 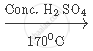
State two used of ethanol (other than as a fuel).
Give the lab. Prepared of methyl alcohol.
What would you see, when ethyne is bubbled through a solution of bromine in carbon tetrachloride?
An organic compound 'P' is a constituent of wine. 'P' on reacting with acidified K2Cr2O7 forms another compound 'Q'. When a piece of sodium is added to 'Q' a gas 'R' evolves which burns with a pop sound. Identify P, Q and R and write the chemical equations of the reactions involved.
Give two uses each of:
(i) Methane (ii) ethane
Name the following:
100% ethanol
State how the following conversion can be carried out:
Ethene to ethyl alcohol
State how the following conversion can be carried out:
Ethyl alcohol to ethene
Answer the following question.
Write the chemical formula and name of the compound which is the active ingredient of all alcoholic drinks. List its two uses.
Write the chemical equation and name of the product formed when this compound reacts with -
(i) Sodium metal
(ii) hot concentrated sulphuric acid
Draw the structures for ethanol.
At room temperature ethanol is _______.
An organic compound ‘A’ is widely used as a preservative and has the molecular formula C2H4O2. This compound reacts with ethanol to form a sweet-smelling compound ‘B’.
Name the process.
______ react with dil. HNO3 to evolve hydrogen gas.
Name the compound formed when ethanol is heated at 443 K in the presence of conc. \[\ce{H2SO4}\] and draw its electron dot structure.
State the role of conc. \[\ce{H2SO4}\] in the reaction.
Give the balanced chemical equation of the following reaction:
Oxidation of ethanol by acidified potassium dichromate.
