Advertisements
Advertisements
प्रश्न
You are given a test tube with two outlets and a bottle of lime water. Using same, how would you demonstrate experimentally the presence of carbon dioxide in air.
उत्तर
Procedure:
- Take lime water in a test tube with two outlets ‘A’ and ‘B’ as shown above.
- Air is sucked slowly through the outlet ‘B’.
- Air moves in through end ‘A’ and passes through the lime water
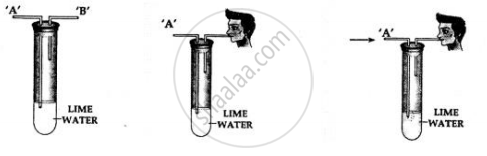
Observation:
The line water in the test tube turns milky.
Conclusion:
Air which is sucked in from and ‘A’ contains carbon dioxide which turns the lime water milky. Hence, air contains – carbon dioxide.
संबंधित प्रश्न
Name the main components of air.
Briefly state the importance of Oxygen.
Give reason:
Mountaineers and divers carry oxygen cylinders with them.
The constituent of air that supports combustion is _____.
How do plants and animals maintain the balance of oxygen and carbon dioxide in the air?
Can you guess why fire extinguishers throw a stream of carbon-di-oxide while putting - off the fire?
Which of the following is true about oxygen?
Nitrogen is essential for burning.
A gas that neither burns nor supports burning is ______.
Boojho took an empty plastic bottle, turned it upside down, and dipped its open mouth into a bucket filled with water. He then tilted the bottle slightly and made the following observations.
(i) Bubbles of air came out from the bottle.
(ii) Some water entered the bottle.
(iii) Nitrogen gas came out in the form of bubbles and oxygen got dissolved in water.
(iv) No bubbles formed, only water entered the bottle.
Which observations is/are correct?
