Advertisements
Advertisements
Question
You are given a test tube with two outlets and a bottle of lime water. Using same, how would you demonstrate experimentally the presence of carbon dioxide in air.
Solution
Procedure:
- Take lime water in a test tube with two outlets ‘A’ and ‘B’ as shown above.
- Air is sucked slowly through the outlet ‘B’.
- Air moves in through end ‘A’ and passes through the lime water
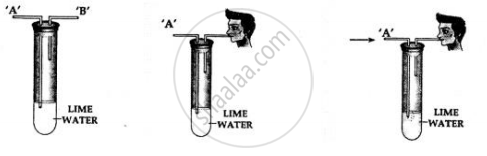
Observation:
The line water in the test tube turns milky.
Conclusion:
Air which is sucked in from and ‘A’ contains carbon dioxide which turns the lime water milky. Hence, air contains – carbon dioxide.
RELATED QUESTIONS
Answer the question put against the following constituents of air:
Nitrogen : Explain its significance for plants and animals.
Define the term Global warming.
Suggest five measures to prevent air pollution.
What are the two ways in which nitrogen fixation occurs?
What do you observe when Calcium is heated and then tested with moist blue and red litmus – paper?
Describe a simple experiment to show that l/5th of air is oxygen.
Name and briefly explain four ways by which pollution of air caused by humans is minimised.
Classify Carbon monoxide. Also give a balanced equation to prepare it.
Write ‘True’ or ‘False’ in front of following statement.
The catalyst used in laboratory preparation of oxygen gas from hydrogen peroxide gas is magnesium oxide
Why is carbon-dioxide gas used to extinguish fire?
