Advertisements
Advertisements
Question
With the help of a labeled diagram, describe a simple experiment to show the presence of oxygen and nitrogen in air using a piece of white phosphorus.
Solution
Activity to show the presence of oxygen and nitrogen in the air using a piece of white phosphorous
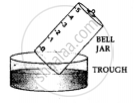
1. A trough is taken and filled with water
2. Over it is placed a bell-jar marked with five equal parts.
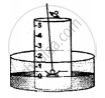
3. A crucible containing white phosphorus is placed on a cork
4. The cork is made to float on the water.
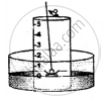
5. A heated iron rod is them taken.
6. The phosphorus is ignited with the heated rod.
Observations:
Dense white fumes of phosphorus pentoxide (P2O5) are formed when the phosphorus (P) burns in the active component of air (oxygen).
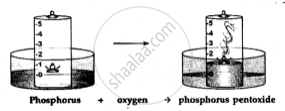
The rise in the level of water in the bell-jar is by 1/5
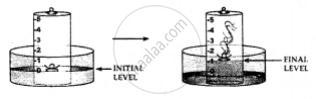
Conclusion:
|
Oxygen (active compound) |
1/5 (approx volume) | Supports combustion | Used up in burning |
|
Nitrogen (In active component bell - jar) |
4/5 (approx volume) | Does not support combustion | Remains behind in the combustion |
The active component of air i.e. oxygen (1/5 of air) is used up in burning. The remaining inactive component of air i.e. nitrogen (4/ 5 of air) is not used up in burning.
RELATED QUESTIONS
What is air pollution ? What are the harmful effects of sulphur dioxide, nitrogen dioxide and hydrogen sulphide present in the air ?
A fuel when used releases least amount of pollutants in the air.
Statement given below is incorrect. Write the correct statement :
Helium gas at low pressure is used for filling electric bulbs.
Classify Calcium oxide. Also give a balanced equation to prepare it.
State the utility of oxygen for respiration in plants
_____ gas turns lime water milky.
______ present in the air is needed for respiration.
Match the following
| 1. | Oxygen | a. | Carbon dioxide |
| 2. | Azote | b. | Nitrogen |
| 3. | Solvay process | c. | Vital life |
| 4. | Gun powder | d. | No life |
Match the following
| 1. | Nitrogen | a. | Acid rain |
| 2. | CO2 | b. | Global warming |
| 3. | Melting of glaciers | c. | Volcanic gases |
| 4. | Corrosion of bridges | d. | Aerated drinks |
Nitrogen gas is so cold that moisture in the air condenses on it, creating a dense fog.
