HSC Science (General)
HSC Science (Electronics)
HSC Science (Computer Science)
Academic Year: 2016-2017
Date & Time: 8th March 2017, 11:00 am
Duration: 3h
Advertisements
An antifriction alloy made up of antimony with tin and copper, which is extensively used in machine bearings is called _______.
(A) Duralumin
(B) Babbitt metal
(C) Spiegeleisen
(D) Amalgam
Chapter: [8.01] D-block Elements
Which of the following pairs is an intensive property?
(A) Density, viscosity
(B) Surface tension, mass
(C) Viscosity, internal energy
(D) Heat capacity, volume
Chapter: [0.03] Chemical Thermodynamics and Energetic
Fe2+ ions react with nitric oxide formed from reduction of nitrate and yields a brown coloured complex _____________.
[Fe(CO)5NO]2+
[Fe(NH3)5NO]2+
[Fe(CH3NH2)5NO]2+
[Fe(H2O)5NO]2+
Chapter: [7.01] Group 15 Elements
MnO2 and Ca3(PO4)2 present in iron ore get reduced to Mn and P in the zone of _______.
(A) combustion
(B) reduction
(C) fusion
(D) slag formation
Chapter: [0.06] General Principles and Processes of Isolation of Elements
An ionic compound crystallises in FCC type structure with ‘A’ ions at the centre of each face and ‘B’ ions occupying corners of the cube. The formula of compound is _______.
AB4
A3B
AB
AB3
Chapter: [0.01] Solid State [0.01] Solid State
On passing 1.5 F charge, the number of moles of aluminium deposited at cathode are _______ [Molar mass of Al = 27 gram mol–1]
(A) 1.0
(B) 13.5
(C) 0.50
(D) 0.75
Chapter: [0.04] Electrochemistry
For a chemical reaction, A→ products, the rate of reaction doubles when the concentration of
‘A’ is increased by a factor of 4, the order of reaction is
(A) 2
(B) 0.5
(C) 4
(D) 1
Chapter: [0.03] Chemical Thermodynamics and Energetic
Write cathode and anode reaction in a fuel cell.
Chapter: [0.04] Electrochemistry [0.05] Electrochemistry
Derive the relation between half life and rate constant for a first order reaction
Chapter: [0.05] Chemical Kinetics [0.06] Chemical Kinetics
Explain magnetic separation process of ores with the help of a neat, labelled diagram.
Chapter: [0.06] General Principles and Processes of Isolation of Elements
Derive the relationship between relative lowering of vapour pressure and molar mass of nonvolatile solute.
Chapter: [0.02] Solutions and Colligative Properties
Define the term ‘enthalpy’.
Chapter: [0.03] Chemical Thermodynamics and Energetic
What will happen to the internal energy if work is done by the system?
Chapter: [0.03] Chemical Thermodynamics and Energetic
Account for the following : Nitrogen does not form pentahalide.
Chapter: [7.01] Group 15 Elements
Calculate the percentage efficiency of packing in case of simple cubic cell.
Chapter: [0.01] Solid State
Advertisements
Write the electronic configuration of the following elements:
Sulphur (Z = 16)
Chapter: [7.02] Group 16 Elements
Write the electronic configuration of the following element:
Krypton (Z = 36)
Chapter: [0.07] Elements of Groups 16, 17 and 18 [7.04] Group 18 Elements
How is phosphine prepared using the following reagent ?
HCl
Chapter: [7.01] Group 15 Elements
How is phosphine prepared using the following reagent?
H2SO4
Chapter: [7.01] Group 15 Elements
How is phosphine prepared using the following reagent?
Caustic soda
Chapter: [7.01] Group 15 Elements
0.05 M NaOH solution offered a resistance of 31.6 Ω in a conductivity cell at 298 K. If the cell constant of the cell is 0.367 cm-1, calculate the molar conductivity of NaOH solution.
Chapter: [0.05] Electrochemistry
Calculate ΔH° for the reaction between ethene and water to form ethyl alcohol from the
following data:
ΔcH° C2H5OH(l) = -1368 kJ
ΔcH° C2H4(g) = -1410 kJ
Does the calculated ΔH° represent the enthalpy of formation of liquid ethanol?
Chapter: [0.03] Chemical Thermodynamics and Energetic
In the Arrhenius equation for a first order reaction, the values of ‘A’ of ‘Ea’ are 4 x 1013 sec-1 and 98.6 kJ mol-1 respectively. At what temperature will its half life period be 10 minutes?
[R = 8.314 J K-1 mol-1]
Chapter: [0.05] Chemical Kinetics
State the first law of electrolysis
Chapter: [0.04] Electrochemistry
Write any two uses of H2SO4
Chapter: [0.04] Electrochemistry
Write any two uses of Chlorine.
Chapter: [0.07] Elements of Groups 16, 17 and 18 [7.03] Group 17 Elements
Distinguish between crystalline solid and amorphous solid
Chapter: [0.01] Solid State
A solution of a substance having mass 1.8 x 10-3 kg has the osmotic pressure of 0.52 atm at 280 K. Calculate the molar mass of the substance used.
[Volume = 1 dm3, R = 8.314 J K-1 mol-1]
Chapter: [0.02] Solutions and Colligative Properties
Define Leaching.
Chapter: [0.06] General Principles and Processes of Isolation of Elements
Define Metallurgy
Chapter: [0.08] Transition and Inner Transition Elements
Define Anisotropy.
Chapter: [0.01] Solid State [0.01] Solid State
Derive an expression for maximum work in isothermal reversible expansion of two moles of an ideal gas.
Chapter: [0.03] Chemical Thermodynamics and Energetic
The boiling point of benzene is 353.23 K. When 1.80 gram of non-volatile solute was dissolved in 90 gram of benzene, the boiling point is raised to 354.11 K. Calculate the molar mass of solute.
[Kb for benzene = 2.53 K kg mol-1]
Chapter: [0.02] Solutions and Colligative Properties
When primary amine reacts with CHCl3 in alcoholic KOH, the product is _______.
(A) aldehyde
(B) alcohol
(C) cyanide
(D) an isocyanide
Chapter: [13.01] Amines
`CH_3-CH_2-Br"Alcoholic KOH"/""> B " HBR"/"">C"Na/Eather"/"">D`, the Compound D is
(A) ethane
(B) propane
(C) n-butane
(D) n-pentane
Chapter: [11.01] Alcohols
Cisplatin compound is used in the treatment of _______.
(A) malaria
(B) cancer
(C) AIDS
(D) yellow fever
Chapter: [16.01] Chemicals in Medicines
A gas when passed through K2Cr2O7 and dil. H2SO4 solution turns it green, the gas is
(A) CO2
(B) NH3
(C) SO2
(D) Cl2
Chapter: [10.01] Haloalkanes [10.02] Haloarenes
The alcohol used in thermometers is _______.
(A) methanol
(B) ethanol
(C) propanol
(D) butanol
Chapter: [11.01] Alcohols
Which of the following vitamins is the vitamin of alicyclic series?
(A) Vitamin C
(B) Vitamin K
(C) Vitamin B
(D) Vitamin A
Chapter: [14.03] Vitamins
Advertisements
Which of the following is the first oxidation product of secondary alcohol?
(A) Alkene
(B) Aldehyde
(C) Ketone
(D) Carboxylic acid
Chapter: [11.01] Alcohols
How is diethyl ether prepared by continuous etherification process?
Chapter: [11.03] Ethers
Write the chemical equation involved in the following reaction:
Hoffmann-bromamide degradation reaction
Chapter: [0.13] Amines [13.01] Amines
How is ethanoic acid prepared from dry ice?
Chapter: [10.01] Haloalkanes [10.02] Haloarenes
Write the molecular and structural formula of BHA and BHT.
Chapter: [11.02] Phenols
Describe laboratory method for preparation of glucose.
Chapter: [14.01] Carbohydrates
Write the factors which are related to the colour of transition metal ions.
Chapter: [8.01] D-block Elements
Explain the given terms: Homopolymers
Chapter: [0.15] Polymers
Explain the given terms : Elastomers
Chapter: [0.15] Polymers
Give IUPAC name of :
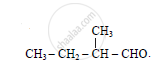
Chapter: [12.01] Aldehydes and Ketones
What is ‘effective atomic number’ (EAN)?
Chapter: [0.09] Coordination Compounds [0.09] Coordination Compounds
Calculate the effective atomic number of the central metal atom in the following compounds:
K4Fe(CN)6 Fe(Z=26)
Chapter: [0.09] Coordination Compounds [0.09] Coordination Compounds
Calculate the effective atomic number of the central metal atom in the following compounds:
Cr(CO)6 Cr(Z=24)
Chapter: [0.09] Coordination Compounds [0.09] Coordination Compounds
Write the different oxidation states of iron
Chapter: [8.02] F-block Elements
Why +2 oxidation state of manganese is more stable?
Chapter: [8.01] D-block Elements
Write a note on ‘aldol condensation’.
Chapter: [0.12] Aldehydes, Ketones and Carboxylic Acids [12.01] Aldehydes and Ketones
Mention two important functions of nucleic acids.
Chapter: [14.04] Nucleic Acids
Mention 'two' functions of lipids
Chapter: [14.02] Proteins
What will be the action of the mixture of sodium nitrite and dilute hydrochloric acid on ethyl amine
Chapter: [13.01] Amines
What will be the action of the mixture of sodium nitrite and dilute hydrochloric acid on aniline
Chapter: [13.01] Amines
What will be the action of the mixture of sodium nitrite and dilute hydrochloric acid on diethyl amine
Chapter: [13.01] Amines
What are ‘antacids’?
Chapter: [16.01] Chemicals in Medicines
Write any ‘two’ side effects of tranquilizers.
Chapter: [16.01] Chemicals in Medicines
Explain the mechanism of alkaline hydrolysis of tert-butyl bromide with energy profile diagram.
Chapter: [12.01] Aldehydes and Ketones
How carbolic acid is prepared from benzene sulphonic acid ?
Chapter: [11.02] Phenols
Other Solutions
Submit Question Paper
Help us maintain new question papers on Shaalaa.com, so we can continue to help studentsonly jpg, png and pdf files
Maharashtra State Board previous year question papers 12th Standard Board Exam Chemistry with solutions 2016 - 2017
Previous year Question paper for Maharashtra State Board 12th Standard Board Exam Chemistry-2017 is solved by experts. Solved question papers gives you the chance to check yourself after your mock test.
By referring the question paper Solutions for Chemistry, you can scale your preparation level and work on your weak areas. It will also help the candidates in developing the time-management skills. Practice makes perfect, and there is no better way to practice than to attempt previous year question paper solutions of Maharashtra State Board 12th Standard Board Exam.
How Maharashtra State Board 12th Standard Board Exam Question Paper solutions Help Students ?
• Question paper solutions for Chemistry will helps students to prepare for exam.
• Question paper with answer will boost students confidence in exam time and also give you an idea About the important questions and topics to be prepared for the board exam.
• For finding solution of question papers no need to refer so multiple sources like textbook or guides.
