SSC (English Medium)
SSC (Marathi Semi-English)
Academic Year: 2019-2020
Date & Time: 16th March 2020, 11:00 am
Duration: 2h
Advertisements
- All questions are compulsory.
- Use of a calculator is not allowed.
- The numbers to the right of the questions indicate full marks.
- In case of MCQs (Q. No. 1(A)) only the first attempt will be evaluated and will be given credit.
- For each MCQ, the correct alternative (A), (B), (C), (D) with sub-question number is to be written as an answer.
For Eg: (i) (A), (ii) (B), (iii) (C) - Scientifically correct, labelled diagrams should be drawn wherever necessary.
According to Mendeleev’s periodic law, properties of elements are periodic function of their _______.
Atomic numbers
Atomic masses
Densities
Boiling points
Chapter: [0.02] Periodic Classification of Elements [0.11] School of Elements
The vapour content in the air is measured using a physical quantity called ____________.
Absolute humidity
Relative humidity
Dew point
Humidity
Chapter: [0.05] Heat
For the normal human eye, the near point is at ___________ cm.
10
20
25
30
Chapter: [0.07] Lenses [0.16] Wonders of Light 1
The astronomical object closest to us is _________ in our galaxy.
Mars
Venus
Jupiter
Moon
Chapter: [0.1] Space Missions
In the Wilfley table method, the particles of gangue are separated by __________ separation method.
Magnetic
Froth floatation
Hydraulic
Gravitational
Chapter:
Tell the odd one out. Give a proper explanation.
Voltmeter
Ammeter
Galvanometer
Thermometer
Chapter: [0.14] The Electric Spark
Complete the correlation:
Alkene : C = C :: Alkyne: _______.
Chapter: [0.09] Carbon Compounds
State true or false:
The frequency of AC is 50 Hz.
True
False
Chapter: [0.15] All about Electromagnetism
Match the Columns:
| Column ‘A’ | Column ‘B’ |
| The wavelength of red light | (a) 600 nm |
| (b) 700 nm | |
| (c) 500 nm |
Chapter: [0.06] Refraction of Light
Advertisements
Name the first artificial satellite sent by Russia in space.
Chapter: [0.1] Space Missions
The weight of an object changes from place to place through its mass is constant.
Chapter:
Write scientific reason.
Stars twinkle but we do not see the twinkling of planets.
Chapter:
Write scientific reason.
Elements belonging to the same group have the same valency.
Chapter: [0.02] Periodic Classification of Elements
How much heat energy is necessary to raise the temperature of 5 kg of water from 20°C to 100°C?
Chapter: [0.05] Heat
Observe the given figure of Fleming’s Right Hand Rule and write the labels of A and B correctly.
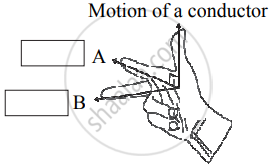
Chapter: [0.04] Effects of Electric Current [0.15] All about Electromagnetism
Observe the given graph and answer the following questions:
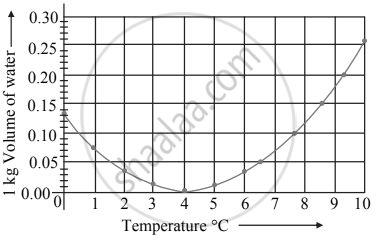
- Name the process represented in the figure.
- At what temperature does this process take place?
Chapter:
- Complete the given chemical reaction:
\[\ce{CuSO4_{(aq)} + Fe_{(s)} ->}\] _______ + _______ - Name the type of the reaction.
Chapter: [0.03] Chemical Reactions and Equations [0.12] The Magic of Chemical Reactions
Write a short note on Alloying.
Chapter: [0.03] Chemical Reactions and Equations [0.08] Metallurgy
Advertisements
An element has its electron configuration as 2, 8, 2. Now answer the following question.
What is the atomic number of this element?
Chapter: [0.02] Periodic Classification of Elements
An element has its electron configuration as 2, 8, 2. Now answer the following question.
What is the group of this element?
Chapter: [0.02] Periodic Classification of Elements
An element has its electron configuration as 2, 8, 2. Now answer the following question.
To which period does this element belong?
Chapter: [0.02] Periodic Classification of Elements
Observe the given figure showing the orbit of a planet moving around the Sun and write the three laws related to it:

The orbit of a planet moving around the Sun
Chapter: [0.01] Gravitation
Read the given passage and answer the following questions:
The home electrical connection consists of ‘live’, ‘neutral’ and ‘earth’ wires. The ‘live’ and the ‘neutral’ wires have potential difference of 220 V. The ‘earth’ is connected to ground. Due to a fault in the equipment or if the plastic coating on the ‘live’ and the ‘neutral’ wires gives a way the two wires come in contact with each other and a large current flows through it producing heat. If any inflammable material (such as wood, cloth, plastic, etc.) exists around that place it can catch fire. Therefore a fuse wire is used as a precautionary measure.
- Name the two wires having potential difference of 220 V.
- What is a short circuit?
- Write the function of a fuse.
Chapter: [0.04] Effects of Electric Current
Observe the given figure and answer the following questions:
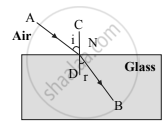
- Name the process represented by the figure.
- State the two laws related to the process.
Chapter:
What is meant by an artificial satellite? How are the satellites classified based on their functions?
Chapter: [0.1] Space Missions
Name two carbon compounds used in day-to-day life.
Chapter:
Observe the given figure of reactivity series of metals and answer the following questions:

Reactivity series of metals
- Name two metals which react with water.
- Name two moderately reactive metals.
- Name the most highly reactive metal and the most less reactive metal.
Chapter: [0.08] Metallurgy
Complete the following table:
| Straight chain of Carbon compounds | Structural formula | Molecular formula | Name |
| C | \[\begin{array}{cc}\ce{H}\\|\\\ce{H - C - H}\\ |\\\ce{H}\end{array}\] |
CH4 | Methane |
| C-C | ______ | ______ | Ethane |
| C-C-C | ______ | C3H8 | ______ |
| C-C-C-C | \[\begin{array}{cc} \ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\\ |\phantom{....}|\phantom{....}|\phantom{....}|\\\ce{H - C - C - C - C - H}\\|\phantom{....}|\phantom{....}|\phantom{....}|\\ \ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H} \end{array}\] |
______ | ______ |
Chapter:
Draw a scientifically correct labelled diagram of a human eye and answer the questions based on it:
- Name the type of lens in the human eye.
- Name the screen at which the maximum amount of incident light is refracted?
- State the nature of the image formed of the object on the screen inside the eye.
Chapter: [0.07] Lenses [0.16] Wonders of Light 1
Observe the following picture and answer the following questions:

- What is rust?
- Write the chemical formula of rust.
- Write the reaction of oxidation of iron at the anode.
- Write the reaction of oxidation of iron at the cathode.
- What is corrosion?
Chapter: [0.03] Chemical Reactions and Equations [0.08] Metallurgy
Other Solutions
Submit Question Paper
Help us maintain new question papers on Shaalaa.com, so we can continue to help studentsonly jpg, png and pdf files
Maharashtra State Board previous year question papers 10th Standard Board Exam Science and Technology 1 with solutions 2019 - 2020
Previous year Question paper for Maharashtra State Board 10th Standard Board Exam -2020 is solved by experts. Solved question papers gives you the chance to check yourself after your mock test.
By referring the question paper Solutions for Science and Technology 1, you can scale your preparation level and work on your weak areas. It will also help the candidates in developing the time-management skills. Practice makes perfect, and there is no better way to practice than to attempt previous year question paper solutions of Maharashtra State Board 10th Standard Board Exam.
How Maharashtra State Board 10th Standard Board Exam Question Paper solutions Help Students ?
• Question paper solutions for Science and Technology 1 will helps students to prepare for exam.
• Question paper with answer will boost students confidence in exam time and also give you an idea About the important questions and topics to be prepared for the board exam.
• For finding solution of question papers no need to refer so multiple sources like textbook or guides.
