Advertisements
Advertisements
Question
A 0.05 M NH4OH solution offers the resistance of 50 ohms to a conductivity cell at 298 K. If the cell constant is 0.50 cm-1 and molar conductance of NH4OH at infinite dilution is 471.4 ohm-1 cm2 mol-1, calculate:
(i) Specific conductance
(ii) Molar conductance
(iii) Degree of dissociation
Solution
Molarity = 0.05 M
Resistance R = 50 ohms
Cell constant, `l/a = 0.50 "cm"^-1`
`A_m^∞ = 471.4 "ohm"^-1 "cm"^2 "mol"^-1`
C = `1/"R" = 1/50 "ohm"^-1`
(i)
Specific conductance,
k = conductance × cell constant
= `1/5 xx 0.50`
= `1/100 = 10^-2 "ohm"^-1 "cm"^-1`
(ii)
Molar conductance,
`A_m^c = (k xx 1000)/("Molarity")`
= `(10^-2 xx 1000)/(0.05)`
= `(10^-2 xx 10^3 xx 10^2)/(5)`
= 200 ohm-1 cm2 mol-1
(iii)
Degree of dissociation,
`α = A_m^c/A_m^∞`
`α = 200/471.4`
= 0.424
APPEARS IN
RELATED QUESTIONS
Fill in the blanks by choosing the appropriate word/words from those given in the brackets:
The unit of conductance is ………. and that of specific conductance is ………..
(Henry’s, aldol condensation, absence, do not, ohm, Raoult’s, increases, common ion effect, easily, three, solubility product, ohm-1, two, four, ohm-1, cm2, Cannizzaro, ohm-1 cm-1, zero, decreases, presence)
The molar conductance of a solution _______ with dilution, while its specific conductance _______ with dilution.
A 0.05 M NH4OH solution offers the resistance of 30.8 ohms to a conductivity cell at 298K. If the cell constant is 0.343 cm−1 and the molar conductance of NH4OH at infinite dilution is 471.4 S cm2 mol−1, calculate the following:
- Specific conductance
- Molar conductance
- Degree of dissociation
In the diagram of the electrolytic cell given below, A, B and C are connected in series having electrolytes of ZnSO4, AgNO3 and CuSO4, respectively.
A steady current of 1.5 A was passed until 1.45 g of Ag was deposited at the cathode of cell B.
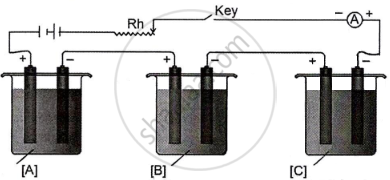
(Atomic mass of Ag = 108, Cu = 63.5, Zn = 65.3)
Answer the following questions:
- How long did the current flow?
- What weight of Cu and Zn was deposited at the cathode?
