Advertisements
Advertisements
Question
A Compound (A) with molecular formula C2H3N on acid hydrolysis gives (B) which reacts with thionyl chloride to give compound (C). Benzene reacts with compound (C) in presence of anhydrous AlCl3 to give compound (D). Compound (D) on reduction with Zn/Hg and Conc. HCl gives (E). Identify (A), (B), (C), (D) and (E). Write the equations.
Solution
1. Compound (A) with molecular formula C2H3N is methyl cyanide. (CH3CN)
2. Methyl cyanide (A) on hydrolysis gives acetic acid (B)
\[\ce{\underset{\underset{(A)}{(Methyl cyanide)}}{CH3CN} ->[H^+/H2O][excess] \underset{\underset{(B)}{(Acetic acid)}}{CH3COOH}}\]
3. Acetic acid (B) with thionyl chloride to give acetyl chloride (C)
\[\ce{\underset{\underset{(B)}{(Acetic acid)}}{CH3COOH} ->[SOCl2] \underset{\underset{(C)}{(Acetyl chloride)}}{CH3COCl} + SO2 + HCl}\]
4. Benzene reacts with acetyl chloride (C) in the presence of anhydrous AlCl3 to give acetophenone (D)

5. Acetyl chloride (C) on reduced in the presence of Pd/H2 and Barium sulphate, to gives acetaldehyde (E).
\[\ce{\underset{\underset{(C)}{(Acetyl chloride)}}{CH3COCl} ->[Pd/H2][BaSO4] \underset{\underset{(E)}{(Acetyldehyde)}}{CH3CHO} + HCl}\]
| A | CH3CN | Methyl cyanide |
| B | CH3COOH | Acetic acid |
| C | CH3COCl | Acetyl chloride |
| D | 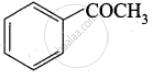 |
Acetophenone |
| E | CH3CHO | Acetaldehyde |
APPEARS IN
RELATED QUESTIONS
Arrange the following in the increasing order of their boiling points: CH3CHO, CH3COOH, CH3CH2OH
Arrange the following compounds in increasing order of their boiling points.
CH3CHO, CH3CH2OH, CH3OCH3, CH3CH2CH3
In the Wolff-Kishner reduction of alkyl aryl ketones, ketones are first converted into ____________.
Which of the following is a solid trimer of formaldehyde?
Identify A in the following reaction.
\[\ce{A + CH3MgBr ->[ether] complex ->[H3O+] (CH3)3C - OH}\]
The reactant (X) in the reaction is:
\[\ce{(X) ->[CH3COONa][(CH3CO)2O] Cinnamic acid}\]
Why is the boiling point of aldehydes and ketones lower than that of corresponding carboxylic acids?
Give a chemical test to distinguish between ethanal acid and ethanoic acid.
