Advertisements
Advertisements
Question
A compound 'X' consists of 4.8% of C and 95.2% of Br by mass.
Name the type of chemical reaction by which X can be prepared from ethane.
Solution
This substance can be prepared by substitution method.
APPEARS IN
RELATED QUESTIONS
Give balanced chemical equations for the following conversions A, B, and C:
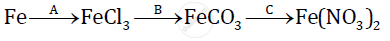
Aluminium carbide reacts with water according to the following equation :
`Al_4C_3 + 12H_2O-> 4Al(OH)_3 + 3CH_4`
1)What mass of aluminium hydroxide is formed from 12g of aluminium carbide?
2) What volume of methane at s.t.p. is obtained from 12g of aluminium carbide?
[Relatively molecular weight of `Al_4Cl_3 = 144; Al(OH)_3 = 78]`
Calculate the relative molecular mass of Sodium acetate
(use K = 39, Cl = 35.5, O = 16, C = 12, H = 1, Na = 23, N = 14, S= 32)
Calculate the relative molecular mass of Ammonium sulphate.
(use K = 39, Cl = 35.5, O = 16, C = 12, H = 1, Na = 23, N = 14, S= 32)
Give one word or phrase for the following:
The ratio of the mass of a certain volume of gas to the mass of an equal volume of hydrogen under the same conditions of temperature and pressure.
Give two tests of the following:
Oxygen
Calculate the relative molecular mass of:
Potassium chlorate
Calculate the relative molecular mass of:
(NH4)2 Cr2O7
Which of the following would weigh most?
What do you understand by the statement that 'vapour density of carbon dioxide is 22'?
