Advertisements
Advertisements
Question
A gas is evolved when ethanol reacts with sodium. Name the gas evolved and also write the balanced chemical equation of the reaction involved.
Solution
Hydrogen gas is evolved when ethanol reacts with sodium.
`2"Na" + 2"C"_2"H"_5"OH" -> underset("Sodium ethoxide")(2"C"_2"H"_5"O"^- "Na"^+) + underset("Hydrogen")("H"_2)`
APPEARS IN
RELATED QUESTIONS
Explain the action of phosphorous trichloride with ethanol. Write the balanced chemical equation of the above reaction.
Write the molecular formula of the 2nd and the 3rd member of the homologous series whose first member is methane.
Write a balanced chemical equation for Burning of ethane in the plentiful supply of air.
Organic compounds having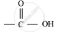 functional group are known as
functional group are known as
What is the molecular formula and structure of the alcohol which can be thought to be derived from pentane?
Name the addition product formed between ethene and water.
How is methane gas prepared in laboratory?
Choose the correct alternative and rewrite the following:
To observe the hydro and clearly, Raju should see it first under the low power lens and then under the high power lens in order to see _____________
Sodium is stored under kerosene. Give reason.
How is ethene prepared from ethanol? Give the reaction involved in it.
