Advertisements
Advertisements
Question
- Give only one suitable chemical test to identify the following gases.
- Ammonia
- Sulphur dioxide
- Hydrogen chloride
- Chlorine
- Carbon dioxide
- Oxygen
- Hydrogen
- Select a basic gas mentioned in Q. (a). How is the basic nature suspected?
- Select acidic gases from the gases mentioned in Q. (a). How is the acidic nature suspected?
- The two gases A and B are bleaching agents. A is greenish-yellow and bleaches due to its oxidizing property while B is a colourless gas that bleaches due to reduction. Identify A and B?
- Which gas turn blue cobalt chloride paper light pink?
Give one similarity in the test between- Cl2 and HCl
- SO2 and CO2.
Solution
-
- Ammonia: If a rod dipped in concentrated hydrochloric acid is brought near ammonia gas, dense white fumes of ammonium chloride (NH4Cl) are formed.
\[\ce{NH3 + HCl -> \underset{Dense white fumes}{NH4Cl}}\] - Sulphur dioxide: It decolorises pink-coloured potassium permanganate solution.
\[\ce{2KMnO4 + 2H2O + 5SO2 -> K2SO4 + 2MnSO4 + 2H2SO4}\] - Hydrogen chloride: When HCl gas is passed through AgNO3 solution, white precipitates of AgCl are formed, which get dissolved in excess of NH4OH.
\[\ce{AgNO3(aq) + HCl ->AgCl\downarrow + HNO3}\] - Chlorine: It turns moist starch iodide paper (KI + starch solution) blue-black.
- Carbon dioxide: When this gas is passed through lime water, it turns milky due to the formation of white precipitates of CaCO3 and on passing excess of carbon dioxide gas, this milkiness disappears.
\[\ce{Ca(OH)2 + CO2 -> \underset{Milky}{CaCO3} + H2O}\] - Oxygen: This gas is absorbed in a colourless alkaline solution of pyrogallol and turns it dark brown.
- Hydrogen: It is neutral to litmus. It burns with a pop sound and pale blue flame when a burning splint is brought near it. On burning with oxygen, the condensed product turns CaCl2 pink.
- Ammonia: If a rod dipped in concentrated hydrochloric acid is brought near ammonia gas, dense white fumes of ammonium chloride (NH4Cl) are formed.
- Ammonia is a basic gas, and its basic nature is suspected through the litmus paper test because it changes the colour of red litmus paper to blue.
\[\ce{NH3 + H2O -> NH4OH}\] - Chlorine, carbon dioxide, hydrogen chloride, hydrogen sulphide, and sulphur dioxide are acidic gases since they convert blue litmus to red.
- A is chlorine and B is sulphur dioxide.
- Light pink water vapour forms on cobalt chloride paper when hydrogen combines with oxygen and condenses.
- A white ppt is created when the gas is passed through a silver nitrate solution.
- The gas turns milky when it passes through the lime water.
APPEARS IN
RELATED QUESTIONS
Give reason for the following:
silver nitrate solution can be used to distinguish HCl from HNO3
Complete and balance the following reaction, state whether dilute or conc. acid is used.
\[\ce{NaHSO3 + HCl ->}\]
Complete the following reaction and balance them.
Ca(HCO3)2 + 2HCl →
Name the gas evolved when dilute hydrochloric acid is added to: Zinc Metal
Convert Hydrochloric acid to nascent chlorine.
Convert Hydrochloric acid to nascent chlorine.
Convert hydrochloric acid to nascent chlorine.
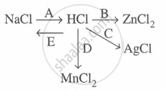
Study the flow chart and give balanced equations with conditions for the conversions A, B, C, D and E.
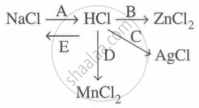
Study the flow chart and give balanced equations with conditions for the conversions A, B, C, D and E.
Complete and balance the following reaction, state whether dilute or conc. acid is used.
\[\ce{NH4OH + HCI->}\]
