Advertisements
Advertisements
Question
A glass tube, sealed at both ends, is 100 cm long. It lies horizontally with the middle 10 cm containing mercury. The two ends of the tube contain air at 27°C and at a pressure 76 cm of mercury. The air column on one side is maintained at 0°C and the other side is maintained at 127°C. Calculate the length of the air column on the cooler side. Neglect the changes in the volume of mercury and of the glass.
Solution
Let CSA of the tube be A.
on the colder side :
P1 = 0.76 m Hg
T1 = 300K
V1 = V
T2 = 273K
V2 = Ax
`(P_1V)/T_1` = `(P_2Ax)/(T_2)`
⇒ `P_2= (P_1 VT_2)/(T_1Ax)`
on the hotter side :
P1= 0.76 m Hg
T1 = 300K
V1' = V
T2' = 400K
V2' = Ay
`(P_1'V)/T_1 = (P_2'Ay)/(T_2')`
⇒ `P_2' = (P_1VT_2')/(T_1Ay)`
In equilibrium , the pressures on both side will balance each other .
⇒ P2' = P2
⇒ `(P_1VT_2')/(T_1Ay) =( P_1VT_2)/(T_1Ax)`
⇒ `(T_2')/y = T_2/x`
From the length of the tube , we get
x + y + 0.1=1
⇒ y = 0.9-x
`400/((0.9-x)) = 273/x`
⇒ x = 0.365 m
⇒ x = 36.5 cm
APPEARS IN
RELATED QUESTIONS
Show that R.M.S. velocity of gas molecules is directly proportional to square root of its absolute temperature.
Prove that root mean square velocity of gas molecule is directly proportional to the square root of its absolute temperature.
At what temperature will average kinetic energy of gas be exactly half of its value at N.T.P.?
If the total kinetic energy per unit volume of gas enclosed in a container is E, the pressure exerted by the gas is
(A) E
(B) 3/2 E
(C) `sqrt3E`
(D) 2/3E
State any four assumptions of kinetic theory of gases.
The quantity \[\frac{pV}{kT}\] represents
There is some liquid in a closed bottle. The amount of liquid is continuously decreasing. The vapour in the remaining part
There is some liquid in a closed bottle. The amount of liquid remains constant as time passes. The vapour in the remaining part
Vapour is injected at a uniform rate in a closed vessel which was initially evacuated. The pressure in the vessel
A vessel A has volume V and a vessel B has volume 2V. Both contain some water which has a constant volume. The pressure in the space above water is pa for vessel A and pbfor vessel B.
Find the average magnitude of linear momentum of a helium molecule in a sample of helium gas at 0°C. Mass of a helium molecule = 6.64 × 10−27 kg and Boltzmann constant = 1.38 × 10−23 J K−1.
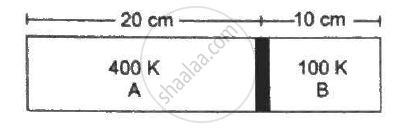
Figure shows a cylindrical tube of length 30 cm which is partitioned by a tight-fitting separator. The separator is very weakly conducting and can freely slide along the tube. Ideal gases are filled in the two parts of the vessel. In the beginning, the temperatures in the parts A and B are 400 K and 100 K respectively. The separator slides to a momentary equilibrium position shown in the figure. Find the final equilibrium position of the separator, reached after a long time.
If the mass of all molecules of a gas is halved and their speeds doubled, then the ratio of initial and final pressures is ________.
Which of the following options may be the correct estimate of the mean free path of gas particles? [n: Number of gas particle per unit volume, d: diameter]
