Advertisements
Advertisements
Question
A metal M forms as oxide having the formula M2O3. It belongs to third period. Write the atomic number and valency of the metal.
Solution
Period no. = no. of shells, so n = 3
From the formula M2O3 its valency is 3.
Since it is a metal, its valence shell has 3 electrons.
So its electronic configuration is 2, 8, 3
Atomic number = 13
Hence the metal is Aluminum with valency 3.
RELATED QUESTIONS
In Period 3 of the Periodic Table, element B is placed to the left of element A. On the basis of this information, choose the correct word from the option to complete the following statement:
The element B would have ______ metallic character than A.
Give the trend in metallic character:
down the group top to bottom.
How does the chemical reactivity of
alkali metals vary?
iii) Observe the figure and answer the following questions.
a) Identify the block shown by box A and write an electronic configuration of any one element of this block.
b) Identify the block of element denoted by letter B and write its period number.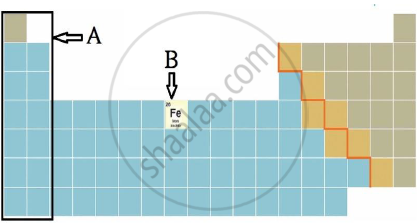
For the main group of the periodic table, the metallic properties of the elements vary approximately with their position as shown in the table.
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 |
| H | He | ||||||
| A | B | ||||||
| C | D |
Will the most non-metallic element be found at A,B,C or D ?
Copy and complete the following sentence choosing the correct word or words from those given below, at the end of the sentence:
The element at the bottom of a group would be expected to show ______ metallic character than the element at the top.
The tendency of an element to form anion is the ______ character of that element.
Silicon is a metallic element.
Write an Explanation.
Electropositivity
The lightest liquid metal is ______
