Advertisements
Advertisements
Question
Write an Explanation.
Electropositivity
Solution
The tendency of an atom of an element to form cation by losing its valence electrons is called electropositivity of that element.
APPEARS IN
RELATED QUESTIONS
Choose the word or phrase from the brackets which correctly complete the following statement:
Metals are good ______ (oxidizing agents/reducing agents) because they are electron ______ (acceptors/donors).
In Period 3 of the Periodic Table, element B is placed to the left of element A. On the basis of this information, choose the correct word from the option to complete the following statement:
The element B would have ______ metallic character than A.
Answer the following in respect of element `31/15 P `
What is its valency?
Give the trend in metallic character:
down the group top to bottom.
Explain the following:
Group 17 elements are strong non-metals, while group 1 elements are strong metals.
Explain the following:
Metallic character of elements decreases from left to right in a period while it increases in moving down a group.
An element X belong to 3rd periods and group II of the periodic table state:
whether it is a metal or a non-metal.
Complete the following sentences
Moving across a ………….. of the periodic table the elements show increasing ………………..character (group, period, metallic, non-metallic).
In Period 3, the most metallic element is ______.
Study the extract of the Periodic Table given below and answer the questions that follow. Give the alphabet corresponding to the element in question. DO NOT repeat an element. 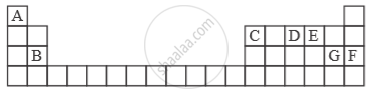
Which non-metallic element has the valency of 2?
Select the correct answer
Configuration of an element is 2, 8, 1. Which of the following statement is true?
Supply the missing word from the words given in brackets.
If an element has one electron in its outermost energy level, then it is likely to be _ (metallic, non metallic)
Copy and complete the following sentence choosing the correct word or words from those given below, at the end of the sentence:
The element at the bottom of a group would be expected to show ______ metallic character than the element at the top.
A group of elements in the periodic table is given below (Boron is the first member of the group and Thallium is the last). Answer the following question in relation to the given group of elements:
Which element has the most metallic character?
The tendency of an element to form anion is the ______ character of that element.
The metal which is hard and has a high melting point and used in filaments of electrical bulbs is ______
Which of the following sets of atomic numbers belong to that of alkali metals?
Which of the following set of elements is written in order of their increasing metallic character?
Choose the odd one out and write the reason:
The given table shows elements with the same number of electrons in its valence shell.
| Elements | A | B | C |
| m.p. | 63.0 | 180.0 | 97.0 |
Arrange them in order of increasing metallic character.
