Advertisements
Advertisements
Question
The given table shows elements with the same number of electrons in its valence shell.
| Elements | A | B | C |
| m.p. | 63.0 | 180.0 | 97.0 |
Arrange them in order of increasing metallic character.
Solution
B < C < A
The metallic character increases as m.p. 180.0 > 97.0 > 63.0 decreases.
RELATED QUESTIONS
Which has the maximum metallic character Na, Li or K?
iii) Observe the figure and answer the following questions.
a) Identify the block shown by box A and write an electronic configuration of any one element of this block.
b) Identify the block of element denoted by letter B and write its period number.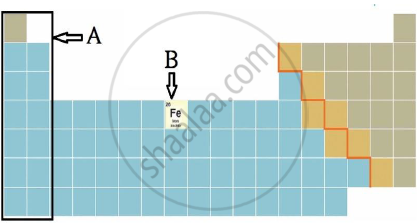
Electronegativity is the metallic property of an element.
Write an Explanation.
Electronegativity
The metal which is hard and has a high melting point and used in filaments of electrical bulbs is ______
The lightest liquid metal is ______
Which of the following sets of atomic numbers belong to that of alkali metals?
Identify the elements with the following property and arrange them in increasing order of their reactivity
- An element which is a soft and reactive metal
- The metal which is an important constituent of limestone
- The metal which exists in liquid state at room temperature
Choose the odd one out and write the reason:
Arrange:
Be, Li, C, B, N, O, F (in increasing metallic character).
