Advertisements
Advertisements
Question
Electronegativity is the metallic property of an element.
Options
Right
Wrong
Solution
Electronegativity is the metallic property of an element- Wrong
APPEARS IN
RELATED QUESTIONS
Answer the following in respect of element `31/15 P `
What is its valency?
Answer the following in respect of element `31/15 P `
Is it a metal or non - metal
Metallic character and non-metallic character are periodic properties discuss ?
Name the periodic property which relates to the character of element which loses one or more electrons when supplied with energy.
How do the following change on moving from left to right in a period of the periodic table?
Give examples in support of your answer.
Nature of oxides of the elements ?
How does the chemical reactivity of
alkali metals vary?
A metal M forms as oxide having the formula M2O3. It belongs to third period. Write the atomic number and valency of the metal.
Choose the correct answer.
An alkaline earth metal
(i) Lead
(ii) potassium
(iii) calcium
(iv) Copper
iii) Observe the figure and answer the following questions.
a) Identify the block shown by box A and write an electronic configuration of any one element of this block.
b) Identify the block of element denoted by letter B and write its period number.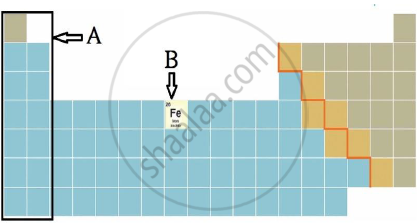
Study the extract of the Periodic Table given below and answer the questions that follow. Give the alphabet corresponding to the element in question. DO NOT repeat an element. 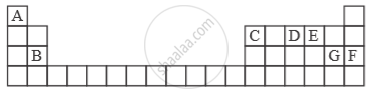
Which non-metallic element has the valency of 2?
Explain
Group 17 elements are non-metals, while group 1 elements are metals.
Within a group, where would you expect to find the element with the greatest metallic character.
A group of elements in the periodic table is given below (Boron is the first member of the group and Thallium is the last). Answer the following question in relation to the given group of elements:
Which element has the most metallic character?
Identify the metallic oxide which is amphoteric in nature.
On moving down the group metallic character increases.
A metal ‘M’ is in the first group of the Periodic Table. What will be the formula of its oxide?
Arrange the following as per instruction given in the bracket.
Cs, Na, Li, K, Rb (increasing metallic character)
Metals are good ______ because they are electron ______.
Arrange:
Be, Li, C, B, N, O, F (in increasing metallic character).
The given table shows elements with the same number of electrons in its valence shell.
| Elements | A | B | C |
| m.p. | 63.0 | 180.0 | 97.0 |
Arrange them in order of increasing metallic character.
