Advertisements
Advertisements
Question
Arrange:
Be, Li, C, B, N, O, F (in increasing metallic character).
Solution
F < O < N < C < B < Be < Li
RELATED QUESTIONS
Is is metal or non-metal?
An element barium has atomic number 56. Look up its position in the periodic table and answer the following question.
Is it a metal or a non-metal?
Study the extract of the Periodic Table given below and answer the questions that follow. Give the alphabet corresponding to the element in question. DO NOT repeat an element. 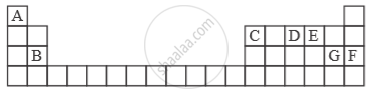
Which non-metallic element has the valency of 2?
State whether the following statement is true or false
All members of zero group are non metals.
Select the correct answer
Which of the following electronic structure is of a metal?
Select the correct answer
Configuration of an element is 2, 8, 1. Which of the following statement is true?
Use the information given in (a) to (h) to identify the substances P to W selecting your answers from the given list.
List:
| Calcium | Oxygen | Copper (II) Oxide |
| Carbon | Calcium hydroxide | Copper (II) Nitrate |
| Lead (II) Oxide | Hydrogen chloride | Chlorine |
| Lead (II) Nitrate | Calcium Oxide | Ammonium chloride |
- P is white solid. When heated produces white fumes (sublime).
- P and R on warming produce an alkaline gas.
- On adding water to T, heat is evolved and R is formed.
- Q burns brightly in the air to form T.
- When S is heated, it gives off brown fumes and leaves a black residue of U.
- A solution of S is formed by warming U with dilute nitric acid.
- V is a gaseous non-metallic element that reacts with hydrogen to form W.
- A solution of W will neutralize the solution of R.
An element has atomic number 17. To which group, the period does it belong? It is metal or non-metal?
Choose the odd one out and write the reason:
Metals are good ______ because they are electron ______.
