Advertisements
Advertisements
प्रश्न
Electronegativity is the metallic property of an element.
पर्याय
Right
Wrong
उत्तर
Electronegativity is the metallic property of an element- Wrong
APPEARS IN
संबंधित प्रश्न
Arrange the following as per the instructions given in the brackets:
Cl, F, Br, I (increasing order of electron affinity)
Answer the following in respect of element `31/15 P `
Is it a metal or non - metal
Metallic character and non-metallic character are periodic properties discuss ?
Give the trend in metallic character:
down the group top to bottom.
Explain the following:
Group 17 elements are strong non-metals, while group 1 elements are strong metals.
The electronic configuration of an element T is 2, 8, 8, 1.
Is it a metal or a non-metal?
Complete the following sentences
Moving across a ………….. of the periodic table the elements show increasing ………………..character (group, period, metallic, non-metallic).
Which has the maximum metallic character Na, Li or K?
iii) Observe the figure and answer the following questions.
a) Identify the block shown by box A and write an electronic configuration of any one element of this block.
b) Identify the block of element denoted by letter B and write its period number.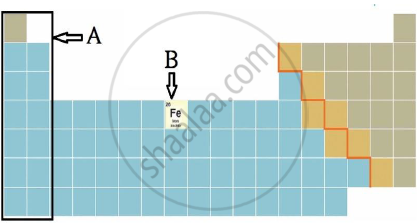
Explain
Larger the atomic size, more metallic is the element.
Within a group, where would you expect to find the element with the greatest metallic character.
Identify the metallic oxide which is amphoteric in nature.
The tendency of an element to form cation is the _______ character of that element.
Write an Explanation.
Electropositivity
3, 1, 2 electrons are in valence shells of X, Y, Z elements. From this information, state the group in which they belong and write their valencies.
An element has atomic number 17. To which group, the period does it belong? It is metal or non-metal?
Which of the following set of elements is written in order of their increasing metallic character?
Identify the elements with the following property and arrange them in increasing order of their reactivity
- An element which is a soft and reactive metal
- The metal which is an important constituent of limestone
- The metal which exists in liquid state at room temperature
A group of elements in the periodic table is given below (Boron is the first member of the group and Thallium is the last).
Boron, Aluminium, Gallium, Indium, Thallium
Answer the following question in relation to the above group of elements:
Will the elements in the group to the right of this boron group be more metallic or less metallic in character? Justify your answer.
