Advertisements
Advertisements
Question
A particle moves in a closed orbit around the origin, due to a force which is directed towards the origin. The de Broglie wavelength of the particle varies cyclically between two values λ1, λ2 with λ1 > λ2. Which of the following statement are true?
- The particle could be moving in a circular orbit with origin as centre.
- The particle could be moving in an elliptic orbit with origin as its focus.
- When the de Broglie wavelength is λ1, the particle is nearer the origin than when its value is λ2.
- When the de Broglie wavelength is λ2, the particle is nearer the origin than when its value is λ1.
Options
b and d
a and c
b, c and d
a, c and d
Solution
b and d
Explanation:
According to the question, here given that the de-Broglie wavelength of the particle can be varying cyclically between two values λ1 and λ2, it is possible if particle is moving in an elliptical orbit with origin as its one focus.
As shown in the figure given alongside,
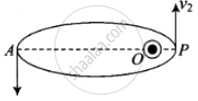
Let v1 and v2 be the speed of particle at A and B respectively and origin is at focus O. If λ1 and λ2 are the de-Broglie wavelengths associated with particle while moving at A and B respectively, then `λ_1 = h/(mv_1)`
And `λ_2 = h/(mv_2)`
∴ `λ_1/λ_2 = v_2/v_1`
Since `λ_1 > λ_2`
∴ `v_2 > v_1`
By the law of conservation of angular momentum, the particle moves faster when it is closer to focus.
From figure, we note that origin O is closed to P than A.
APPEARS IN
RELATED QUESTIONS
What is the
(a) momentum,
(b) speed, and
(c) de Broglie wavelength of an electron with kinetic energy of 120 eV.
The wavelength of light from the spectral emission line of sodium is 589 nm. Find the kinetic energy at which
(a) an electron, and
(b) a neutron, would have the same de Broglie wavelength.
Find the de Broglie wavelength of a neutron, in thermal equilibrium with matter, having an average kinetic energy of `(3/2)` kT at 300 K.
Sodium and copper have work function 2.3 eV and 4.5 eV respectively. Then, the ratio of the wavelengths is nearest to ______.
A proton and α-particle are accelerated through the same potential difference. The ratio of the de-Broglie wavelength λp to that λα is _______.
A proton and an α-particle are accelerated, using the same potential difference. How are the de-Broglie wavelengths λp and λa related to each other?
Assuming an electron is confined to a 1 nm wide region, find the uncertainty in momentum using Heisenberg Uncertainty principle (∆x∆p ≃ h). You can assume the uncertainty in position ∆x as 1 nm. Assuming p ≃ ∆p, find the energy of the electron in electron volts.
Two particles A and B of de Broglie wavelengths λ1 and λ2 combine to form a particle C. The process conserves momentum. Find the de Broglie wavelength of the particle C. (The motion is one dimensional).
Two particles move at a right angle to each other. Their de-Broglie wavelengths are λ1 and λ2 respectively. The particles suffer a perfectly inelastic collision. The de-Broglie wavelength λ, of the final particle, is given by ______.
The ratio of wavelengths of proton and deuteron accelerated by potential Vp and Vd is 1 : `sqrt2`. Then, the ratio of Vp to Vd will be ______.
