Advertisements
Advertisements
Question
A simple U tube contains mercury to the same level in both of its arms. If water is poured to a height of 13.6 cm in one arm, how much will be the rise in mercury level in the other arm?
Given : density of mercury = 13.6 x 103 kg m-3 and density of water = 103 kg m-3.
Solution
Given, ρm = 13.6 × 103 kg m -3, ρw = 103 kg m -3
Height to which water is poured in one arm, hw = 13.6 cm
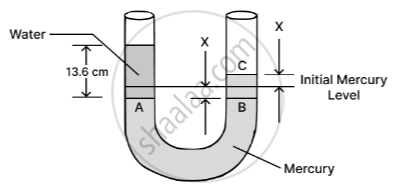
By pouring 13.6 cm of water, the mercury level in the left arm goes down to point A by x cm, while in the right arm, it rises to point C by x cm. Therefore, BC = hm = 2x cm
By Pascal's law,
Pressure in the water column = pressure in the mercury column
Therefore, PA = PB
⇒ hw ρw g = hm ρm g
⇒ 13.6 × 103 × g = 2𝑥 × 13.6 × 103 × g
⇒ 1 = 2𝑥
⇒ 𝑥 = `1/2` = 0.5 cm
Hence, the rise in mercury level = 0.5 cm
APPEARS IN
RELATED QUESTIONS
What is the other name of buoyant force ?
Name the force which makes heavy objects appear light when immersed in a liquid.
A body X sinks in water, but a body Y floats on water. The density of the body X is ......... than the density of body Y.
Distinguish between density and relative density.
Complete the following sentence :
Pressure at all points at the same depth is ....................
The pressure inside a liquid of density p at a depth h is :
A metal solid cylinder tied to a thread is hanging from the hook of a spring balance. The cylinder is gradually immersed into the water contained in a jar. What changes do you expect in the readings of the spring balance? Explain your answer.
A beaker contains a liquid of density ‘ρ’ up to height ‘h’ such that ‘PA’ is atmospheric pressure and ‘g’ is the acceleration due to gravity. Answer the following questions:
- What is the pressure on the free surface of the liquid?
- What is the pressure on the base of the beaker?
- What is the lateral pressure at the base on the inner walls of the beaker?
Select the correct option:
The phenomenon due to which a solid experiences upward force when immersed in water is called:
Consider the statement given below and choose the correct option.
Assertion: Water strider slides easily on the surface of the water.
Reason: Water strider experiences less buoyant force.
