Advertisements
Advertisements
Question
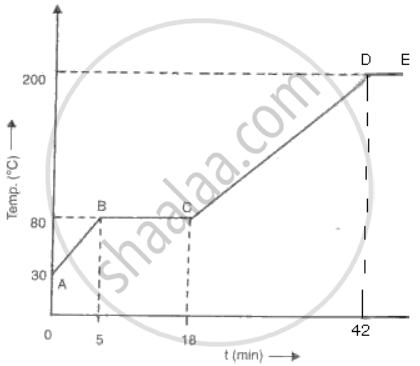
A solid of mass 100 g is heated with a burner which is supplying heat at the rate of 100 J/s. The graph in the following fig shows the changes that take place.
(a) What is the melting point of the substance?
(b) What is boiling point of the substance?
(c) Calculate the Sp. heat cap. of solid.
(d) Calculate the Sp. latent heat of fusion.
(e) Calculate the Sp. heat cap. of the liquid.
Solution
(a) Melting point is 80°C
(b) Boiling point is 200°c.
(c) In 5 min, change in temperature= 50°C
`"Q"/"t"` = 100 J/s
Heat supplied in 5 min,Q = lOOx 30 = 3000J
Q = mCs ΔT
Cs = `"Q"/"m"Δ"T" = 3000/(100 xx 50)` = 0.6 J/g°C
( d) From 5 min tD 18 min, hea: supplied, Q= 780 x 100 = 78000 J
Q=ml
78000= l00 x L
L=780 J/g
( e) From 18 min to 40 min, change in tempera:ure = 120°C
`"Q"/"t"` = l00 J/s
Heat supplied in 22 min, Q = 100 x 1320 = 132000 J
Q= mCLΔT
CL = `"Q"/("m"Δ"T") = 132000/(100 xx 120)` = 11 J / g°C
