Advertisements
Advertisements
Question
Accomplish the following conversion:
Benzene to m-bromophenol
Solution
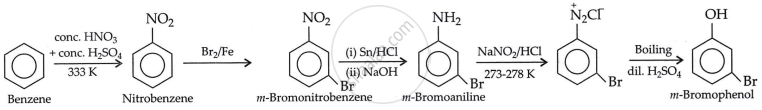
APPEARS IN
RELATED QUESTIONS
Give reasons for the following: (CH3)2NH is more basic than (CH3)3N in an aqueous solution.
How will you convert Benzene into N, N-dimethylaniline?
Arrange the following in increasing order of their basic strength:
CH3NH2, (CH3)2NH, (CH3)3N, C6H5NH2, C6H5CH2NH2
Complete the following acid-base reaction and name the product:
\[\ce{(C2H5)3N + HCl ->}\]
Arrange the following:
In increasing order of boiling point:
C2H5OH, (CH3)2NH, C2H5NH2
Accomplish the following conversions - Aniline to p-bromoaniline
Give plausible explanation for each of the following:
Why are aliphatic amines stronger bases than aromatic amines?
Arrange the following in the increasing order of their pKb values:
C6H5NH2, C2H5NH2, C6H5NHCH3
Arrange the following in increasing order of basic strength :
C6H5NH2, C6H5NHCH3, C6H5N(CH3)2
Answer in one sentence.
Arrange the following amines in increasing order of boiling points.
n-propylamine, ethylmethyl amine, trimethylamine.
Tertiary amines have lowest boiling points because ________________
Assertion: N-Ethylbenzene sulphonamide is soluble in alkali.
Reason: Hydrogen attached to nitrogen in sulphonamide is strongly acidic.
Acetic acid exist as dimer in benzene due to
Which of the following have less melting point than amine?
Which of the following amines form maximum hydrogen bonds within themselves?
Which one of the following compounds will liberate CO2, when treated with NaHCO3?
Arrange the decreasing boiling point.
\[\ce{CH3COOH, C2H5OH, CH3NH2, CH3OCH3}\]
Write short note on the following:
Ammonolysis
