Advertisements
Advertisements
Question
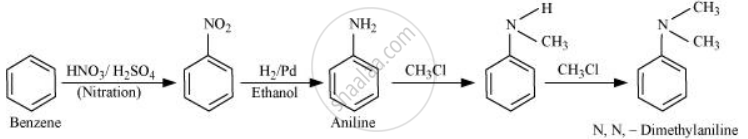
How will you convert Benzene into N, N-dimethylaniline?
Solution
APPEARS IN
RELATED QUESTIONS
Give reasons for the following: (CH3)2NH is more basic than (CH3)3N in an aqueous solution.
Arrange the following in increasing order of their basic strength :

- Write structures of different isomeric amines corresponding to the molecular formula C4H11N.
- Write the IUPAC names of all the isomers.
- What type of isomerism is exhibited by different pairs of amines?
Arrange the following in increasing order of their basic strength:
C2H5NH2, C6H5NH2, NH3, C6H5CH2NH2 and (C2H5)2NH
Complete the following acid-base reaction and name the product:
\[\ce{CH3CH2CH2NH2 + HCl ->}\]
Complete the following acid-base reaction and name the product:
\[\ce{(C2H5)3N + HCl ->}\]
Write a short note on ammonolysis.
Complete the following reactions:
`C_6H_5N_2Cl+H_3PO_2+H_2O->`
Give reasons CH3NH2 is more basic than C6H5NH2.
Tertiary amines have lowest boiling points because ________________
Arrange the following compounds in increasing order of their boiling points.
Ethyl alcohol, Ethyl amine, Ethanoic acid, Ethane
The CORRECT decreasing order of solubility in water will be ____________.
Among the following isomeric amines, an amine having highest boiling point is:
Which of the following should be most volatile?
A compound Z with molecular formula \[\ce{C3H9N}\] reacts with \[\ce{C6H5SO2Cl}\] to give a solid, insoluble in alkali. Identify Z.
Dimerisation of carboxylic acids is due to
Which of the following compound gives a secondary amine oh reduction?
Which of the following amines form maximum hydrogen bonds within themselves?
