Advertisements
Advertisements
Question
Account for the following:
Ethylamine is soluble in water, whereas aniline is not.
Solution
Ethylamine is soluble in water, due to hydrogen bonding as shown below:
\[\begin{array}{cc}
\ce{H}\phantom{..........................}\ce{H}\phantom{.................}\\
|\phantom{...........................}|\phantom{.................}\\
\ce{{- - - - - -} N-H {- - - - - -} O-H {- - - - - -} N-H {- - - - - -} O-H {- - - - - -}}\\
\phantom{..........}|\phantom{.............}|\phantom{..............}|\phantom{.............}|\phantom{.............}\\
\ce{\underset{Ethylamine}{C2H5}}\phantom{.......}\ce{H}\phantom{.............}\ce{C2H5}\phantom{.........}\ce{H}\phantom{.....}
\end{array}\]
On the other hand, aniline has a large phenyl group and has -I effect due to which the hydrogen bonding with water is reduced. Hence, aniline is insoluble in water.
APPEARS IN
RELATED QUESTIONS
Which among the following molecular formulae represents urotropine?
(a) C6H12N4
(b) C6H24H4
(c) C6H12N4O2
(d) C6H24N4O2
Write the IUPAC name of the following compound and classify it as primary, secondary and tertiary amine.
(CH3)3CNH2
Write the IUPAC name of the following compound and classify it as primary, secondary and tertiary amine.
(CH3CH2)2NCH3
Give one chemical test to distinguish between the following pair of compounds.
Aniline and benzylamine
Give one chemical test to distinguish between the following pair of compounds.
Aniline and N-methylaniline
Account for the following:
pKb of aniline is more than that of methylamine.
How will you convert ethanoic acid into methanamine?
How will you convert ethanoic acid into propanoic acid?
How will you convert methanamine into ethanamine?
How will you convert propanoic acid into ethanoic acid?
Accomplish the following conversion:
Chlorobenzene to p-chloroaniline
Complete the following reaction:
\[\ce{C6H5NH2 + H2SO4 (conc.)}\]
Complete the following reaction:
\[\ce{C6H5NH2 + (CH3CO)2O ->}\]
Complete the following reaction:
\[\ce{C6H5N2Cl ->[(i) HBF4][(ii) NaNO2/Cu, \Delta]}\]
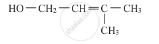
Write the IUPAC name of the given compound :
Give simple chemical tests to distinguish between the following pair of compounds:
Benzaldehyde and Benzoic acid
Using IUPAC norms write the formula of Hexaamminecobalt (III) sulphate.
Do the following conversions in not more than two steps:

