Advertisements
Advertisements
Question
Account for the following :
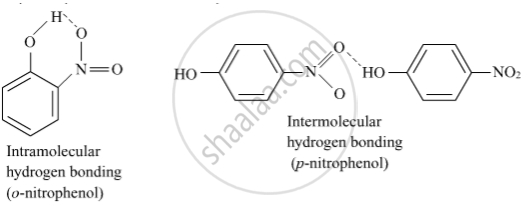
o-nitrophenol is more steam volatile than p-nitrophenol.
Solution
o-Nitrophenol is steam volatile due to intramolecular hydrogen bonding while p- nitrophenol is less volatile due to intermolecular hydrogen bonding
APPEARS IN
RELATED QUESTIONS
An ionic compound crystallises in FCC type structure with ‘A’ ions at the centre of each face and ‘B’ ions occupying corners of the cube. The formula of compound is _______.
Classify the following solid:
Pb
An ionic compound AxBy crystallizes in fcc type crystal structure with B ions at the centre of each face and A ion occupying corners of the cube. the correct formula of AxBy is
Solid CO2 is an example of ____________.
In calcium fluoride, having the flurite structure the coordination number of Ca2+ ion and F− Ion are ____________.
Classify the following solid.
Iodine
Silicon carbide is an example of ____________.
Solid CH4 is ____________.
Graphite cannot be classified as ______.
Why are liquids and gases categorised as fluids?
