Advertisements
Advertisements
Question
Account for the following:
Phenol is a stronger acid than aliphatic alcohol.
Solution
Phenol is more acidic than alcohols due to stabilisation of phenoxide ions through resonance.
In Phenol
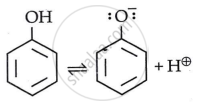

By stabilizing the phenoxide ion, the presence of an electron withdrawing group raises the acidity of phenol, while the presence of an electron releasing group decreases the acidity of phenol.
APPEARS IN
RELATED QUESTIONS
Write the molecular and structural formula of BHA and BHT.
How carbolic acid is prepared from benzene sulphonic acid ?
Give one chemical test to distinguish between the following pair of compounds: Acetone and phenol.
How will you obtain the following? (Give balanced equation.)
Picric acid from phenol.
How will you obtain the following? (Give balanced equation.)
(i) Anisole from phenol
(ii) Ethyl acetate from ethanol.
What is the product A obtained in the following reaction?
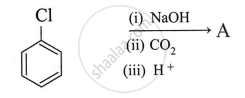
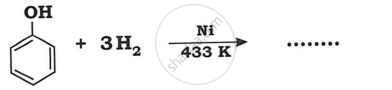
Identify product in:
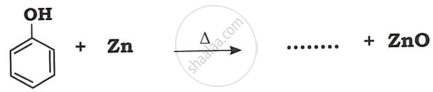
Identify product in:
Match the following:
| (i) | Phenol | (a) | Hexane + heptane |
| (ii) | EDTA | (b) | Globular protein |
| (iii) | Ideal solution | (c) | Azo dye |
| (iv) | Insulin | (d) | Hexadentate ligand |
Write a chemical test to distinguish between ethanol and phenol.
