Advertisements
Advertisements
Question
How will you obtain the following? (Give balanced equation.)
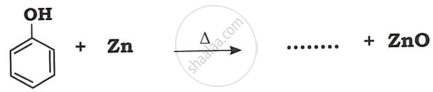
(i) Anisole from phenol
(ii) Ethyl acetate from ethanol.
Solution
(i)
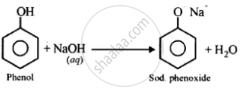
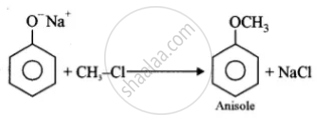
(ii)
\[\ce{\underset{\text{Ethanol}}{C2H5OH } + \underset{\text{Acetic acid}}{CH3COOH}<=>[conc H2SO4,\triangle] \underset{\text{Ethyl acetate}}{CH3COOC2H5}+H2O}\]
APPEARS IN
RELATED QUESTIONS
Write the molecular and structural formula of BHA and BHT.
How carbolic acid is prepared from benzene sulphonic acid ?
Give one chemical test to distinguish between the following pair of compounds: Acetone and phenol.
How will you obtain the following? (Give balanced equation.)
Picric acid from phenol.
What is the product A obtained in the following reaction?
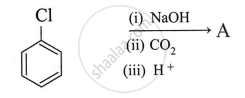
Identify product in:
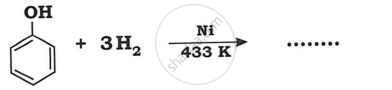
Identify product in:
Match the following:
| (i) | Phenol | (a) | Hexane + heptane |
| (ii) | EDTA | (b) | Globular protein |
| (iii) | Ideal solution | (c) | Azo dye |
| (iv) | Insulin | (d) | Hexadentate ligand |
Account for the following:
Phenol is a stronger acid than aliphatic alcohol.
Write a chemical test to distinguish between ethanol and phenol.
