Advertisements
Advertisements
Questions
How carbolic acid is prepared from benzene sulphonic acid ?
Prepare carbolic acid from benzene sulphonic acid.
Solution
Preparation of Carbolic acid (Phenol) from Benzene sulphonic acid (C6H6SO3H):
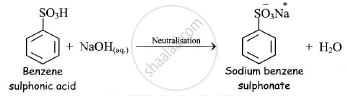
a) When benzene sulphonic acid is neutralised by aqueous sodium hydroxide (NaOH),
sodium benzene sulphonate is obtained.
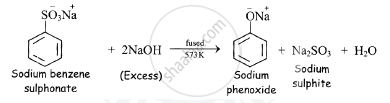
b) Dry sodium benzene sulphonate when fused with excess of sodium hydroxide at 573K,
sodium phenoxide is obtained along with sodium sulphite (Na2SO3).
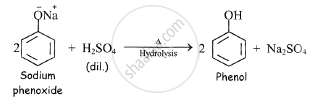
c) sodium phenoxide when hydrolysed by heating with dilute sulphuric acid, phenol is
obtained.
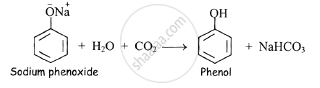
When a current of carbon dioxide is passed through aqueous sodium phenoxide, phenol is obtained as product.
APPEARS IN
RELATED QUESTIONS
Write the molecular and structural formula of BHA and BHT.
Define carbolic acid.
Give one chemical test to distinguish between the following pair of compounds: Acetone and phenol.
How will you obtain the following? (Give balanced equation.)
Picric acid from phenol.
How will you obtain the following? (Give balanced equation.)
(i) Anisole from phenol
(ii) Ethyl acetate from ethanol.
What is the product A obtained in the following reaction?
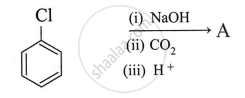
Identify product in:
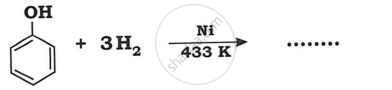
Identify product in:
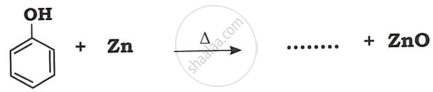
Match the following:
| (i) | Phenol | (a) | Hexane + heptane |
| (ii) | EDTA | (b) | Globular protein |
| (iii) | Ideal solution | (c) | Azo dye |
| (iv) | Insulin | (d) | Hexadentate ligand |
Account for the following:
Phenol is a stronger acid than aliphatic alcohol.
Write a chemical test to distinguish between ethanol and phenol.
