Advertisements
Advertisements
Question
Among the elements of the second period, Li to Ne, pick out the element that is the most reactive metal
Solution
The most reactive metal: Lithium
Concept Insight: Lithium is the most reactive metal as it can complete its octet by losing its single electron present in its outermost shell.
APPEARS IN
RELATED QUESTIONS
Arrange the following as per the instructions given in the brackets:
Na, K, Cl, S, Si (increasing order ionization energy)
In Period 3 of the Periodic Table, element B is placed to the left of element A. On the basis of this information, choose the correct word from the option to complete the following statement:
The element B would have ______ metallic character than A.
Name any five periods properties.
Explain the following:
Group 17 elements are strong non-metals, while group 1 elements are strong metals.
Arrange the elements of group 17 and group 1 according to the given conditions.
Increasing non – metallic character
iii) Observe the figure and answer the following questions.
a) Identify the block shown by box A and write an electronic configuration of any one element of this block.
b) Identify the block of element denoted by letter B and write its period number.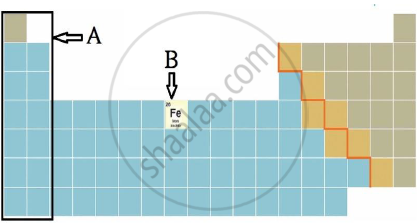
Electronegativity is the metallic property of an element.
Write scientific reason.
The metallic character of elements increases while going down the groups.
Write scientific reason.
The non-metallic character increases while going from left to right in a period.
The lightest liquid metal is ______
