Advertisements
Advertisements
Question
An element X (atomic number 17) reacts with an element Y (atomic number 20) to form a divalent halide.
- Where in the periodic table are elements X and Y placed?
- Classify X and Y as metal (s), non-metal (s) or metalloid (s)
- What will be the nature of oxide of element Y? Identify the nature of bonding in the compound formed
- Draw the electron dot structure of the divalent halide
Solution
- Element X is chlorine, it is placed at 17th group and 3rd period in the periodic table. Element Y is calcium, it is placed at the 2nd group and 4th period in the periodic table.
- Element X is a non-metal and element Y is a metal.
- Element Y is metal and thus, the oxide of this metal will be basic in nature. The chemical formula for calcium oxide is CaO and the bonding in the compound is ionic in nature.
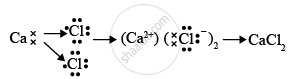
APPEARS IN
RELATED QUESTIONS
Answer the following in respect of element `31/15 P `
Is it a reducing agent or oxidizing agent?
What do you understand by atomic size? State its unit?
Size of atom progressively becomes smaller when we move from sodium (Na) to chlorine (CI) in the third period of the periodic table?
Fill in the blanks.
Size of the atoms ______ from left to right across a period and ______ on descending in a group of normal elements.
The changes in the properties of elements on moving from left to right across a period of the Periodic Table. For the property, choose the correct answer.
The atomic size:
Nanometer unit is used to measure atomic radius.
Write an Explanation.
Atomic radius
Elements have been arranged in the following sequence on the basis of their increasing atomic masses.
| F, | Na, | Mg, | Al, | Si, | P, | S, | Cl, | Ar, | K |
- Pick two sets of elements which have similar properties.
- The given sequence represents which law of classification of elements?
- Electropositive nature of the element(s) increases down the group and decreases across the period
- Electronegativity of the element decreases down the group and increases across the period
- Atomic size increases down the group and decreases across a period (left to right)
- Metallic character increases down the group and decreases across a period.
On the basis of the above trends of the Periodic Table, answer the following about the elements with atomic numbers 3 to 9.
- Name the most electropositive element among them
- Name the most electronegative element
- Name the element with smallest atomic size
- Name the element which is a metalloid
- Name the element which shows maximum valency.
Which one has the largest size?
