Advertisements
Advertisements
प्रश्न
An element X (atomic number 17) reacts with an element Y (atomic number 20) to form a divalent halide.
- Where in the periodic table are elements X and Y placed?
- Classify X and Y as metal (s), non-metal (s) or metalloid (s)
- What will be the nature of oxide of element Y? Identify the nature of bonding in the compound formed
- Draw the electron dot structure of the divalent halide
उत्तर
- Element X is chlorine, it is placed at 17th group and 3rd period in the periodic table. Element Y is calcium, it is placed at the 2nd group and 4th period in the periodic table.
- Element X is a non-metal and element Y is a metal.
- Element Y is metal and thus, the oxide of this metal will be basic in nature. The chemical formula for calcium oxide is CaO and the bonding in the compound is ionic in nature.
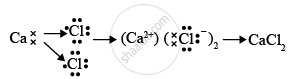
APPEARS IN
संबंधित प्रश्न
Answer the following in respect of element `31/15 P `
Is it a reducing agent or oxidizing agent?
Answer the following in respect of element `31/15 P `
Give its formula with chlorine?
Give the trends in atomic size on moving down the group.
Arrange the following in increasing order of property indicated
I, I+, I- (atomic size)
Select the correct answer
Atomic radii of fluorine and neon in angstrom unit are
Write an Explanation.
Atomic radius
Some elements and their atomic radii are given here. Arrange them in decreasing order of their atomic radii. Identify which of the above elements is the biggest atom and which is smallest?
| Element | K | Na | Rb | Cs | Li |
| Atomic radius (pm) | 231 | 186 | 244 | 262 | 151 |
Elements have been arranged in the following sequence on the basis of their increasing atomic masses.
| F, | Na, | Mg, | Al, | Si, | P, | S, | Cl, | Ar, | K |
- Pick two sets of elements which have similar properties.
- The given sequence represents which law of classification of elements?
An element with the largest atomic radius among the following is ______.
