Advertisements
Advertisements
Question
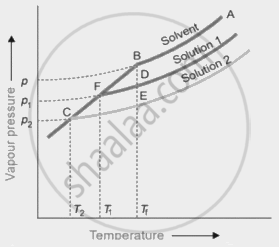
An experiment was carried out in the laboratory to study depression in freezing point. 1 M aqueous solution of \[\ce{Al(NO3)3}\] and 1 M aqueous solution of glucose were taken. From the given figure, identify solution 1 and solution 2. Give a plausible reason for your answer.
Solution
Depression in the freezing point is a colligative property. In dilute solutions, the depression of the freezing point (ΔTf) is directly proportional to the molal concentration of the solute in a solution. From the graph, it is interpreted that Solution 2 shows more depression in the freezing point.
1 M \[\ce{Al(NO)3}\] has a higher i value (i = 3) than 1 M glucose (i = 1).
1 M \[\ce{Al(NO)3}\] will have higher depression; hence, solution 2 is \[\ce{Al(NO)3}\] solution and solution 1 is glucose solution.
