Advertisements
Advertisements
Question
An organic compound on treatment with concentrated NaOH gave 2, 2-dimethylpropan-1-ol and sodium salt of 2, 2-dimethylpropanoic acid. The compound is:
Options
\[\ce{CH3CH2CH2CH2CHO}\]
\[\begin{array}{cc}
\ce{CH3}\phantom{.....}\\
|\phantom{.......}\\
\ce{CH3 - CH - CH2CHO}
\end{array}\]\[\begin{array}{cc}
\phantom{..}\ce{CH3}\\
|\phantom{.}\\
\ce{CH3 - C - CHO}\\
|\phantom{.}\\
\phantom{..}\ce{CH3}
\end{array}\]\[\begin{array}{cc}
\ce{O}\\
||\\
\ce{CH3CH2CCH2CH3}
\end{array}\]
Solution
\[\begin{array}{cc}
\phantom{..}\ce{CH3}\\
|\phantom{.}\\
\ce{CH3 - C - CHO}\\
|\phantom{.}\\
\phantom{..}\ce{CH3}
\end{array}\]
Explanation:
The chemical goes through the Cannizzaro reaction. As a result, there should be no α-hydrogen atom.
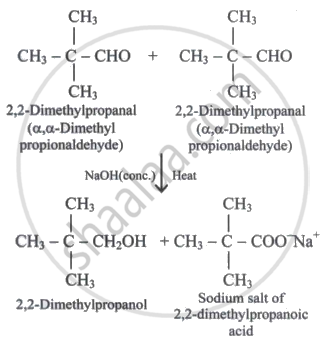
α-H atoms are found in the other three aldehydes. As a result, aldol condensation occurs, resulting in β-hydroxy aldehydes.
