Advertisements
Advertisements
Question
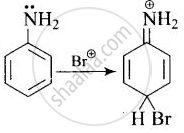
Arenium ion involved in the bromination of aniline is:
(i)
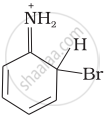
(ii)
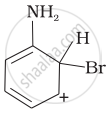
(iii)
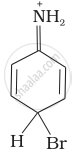
(iv)
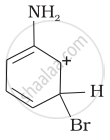
Solution
(i)

(ii)

(iii)

Explanation:
Arenium ion involved in the bromination of aniline are as follows:
(i)
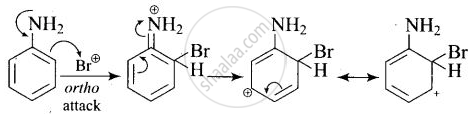
(ii)
APPEARS IN
RELATED QUESTIONS
Illustrate the following reaction giving suitable example in each case: Diazotisation
Write equations of the following reactions:
Acetylation of aniline
Why is aniline soluble in aqueous HCl?
How will you carry out the following conversions?
p-toluidine diazonium chloride `→` p-toluic acid
Match the compounds given in Column I with the items given in Column II.
| Column I | Column II | ||
| (i) | Benzene sulphonyl chloride | (a) | Zwitter ion |
| (ii) | Sulphanilic acid | (b) | Hinsberg reagent |
| (iii) | Alkyl diazonium salts | (c) | Dyes |
| (iv) | Aryl diazonium salts | (d) | Conversion to alcohols |
Aniline when treated with cone. HNO3 gives
Consider the following compounds:
(i) p-methyl aniline
(ii) N, N-dimethylaniline
(iii) N-ethyl aniline
(iv) N-ethyl-N-methyl aniline
The compounds which do not form diazonium salt with ice-colds NaNO2 and HCl are:
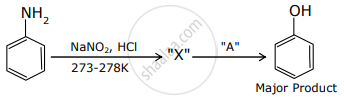
Major Product In the above chemical reaction, intermediate "X" and reagent/condition "A" are:
Aniline dissolved in dilute HCl is reacted with sodium nitrate at 0 °C. This solution was added dropwise to a solution containing equimolar mixture of aniline and phenol in dil. HCl. The structure of the major product is:
How will the following be converted? (Give chemical equation)
Aniline to benzene diazonium chloride.
