Advertisements
Advertisements
Question
Balance the following equation Also name the product formed.
`"K" + "CI"_2 → "KCI"`
Solution
`2"K" + "CI"_2 → 2 "KCI"` Potassium chloride
APPEARS IN
RELATED QUESTIONS
What do you understand by exothermic reactions?
The chemical reaction between quicklime and water is characterised by:
(a) evolution of hydrogen gas
(b) formation of slaked lime precipitate
(c) change in temperature of mixture
(d) change in colour of the product
Write your observations and name the product when
Iron nails are added to an aqueous solution of copper sulphate.
Write the chemical equation for the following word equation and balance them.
Calcium + Nitrogen → Calcium nitride
Balance the following equation:
Pb3O4 → PbO + O2
How is a flame test performed?
A student mixes sodium sulphate powder in barium chloride powder. What change would the student observe on mixing the two powders? Justify your answer and explain how he can obtain the desired change.
Representation of the results of a chemical change – is a chemical equation.
For the equation: FeCl3 + 3NH4OH 3NH4Cl + Fe(OH)3 ↓
Answer the following:
Is the equation given above a balanced equation. Give reasons.
\[\ce{2KClO3->[MnO2] 2KCl + 3O2[g] - is a balanced equation.}\]
State what is a ‘balanced equation’.
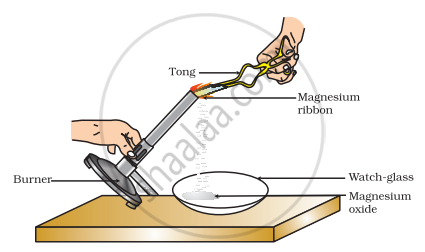
Which of the following is the correct observation of the reaction shown in the above set up?
