Advertisements
Advertisements
Question
Between sodium hydrogen carbonate and magnesium hydroxide which is a better antacid and why?
Solution
Magnesium hydroxide is a better antacid because being insoluble it does not allow the pH to increase above neutral. Hydrogencarbonate being soluble, its excess can make the stomach alkaline and trigger the production of even more acid.
APPEARS IN
RELATED QUESTIONS
What is meant by the term ‘broad spectrum antibiotics’? Explain.
Which of the following statements is not correct.
Which of the following are sulpha drugs?
(i) Sulphapyridine
(ii) Prontosil
(iii) Salvarsan
(iv) Nardil
Which of the following statements are incorrect about penicillin?
(i) An antibacterial fungus.
(ii) Ampicillin is its synthetic modification.
(iii) It has bacteriostatic effect.
(iv) It is a broad-spectrum antibiotic.
What is the mode of action of antimicrobial drugs?
In what respect do prontosil and salvarsan resemble. Is there any resemblance between azo dye and prontsil? Explain.
The mixture of chloroxylenol and terpineol is an example of ______.
How many of the following drugs is/are example(s) of broad spectrum antibiotic?
The structure shown below is of which well-known drug molecule-
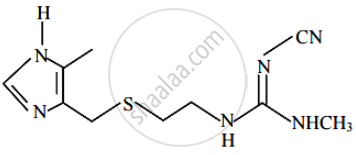
Sulphonamides act as ______.
