Advertisements
Advertisements
Question
Calculate magnetic moment of thorium (Z=90). Is this element diamagnetic or paramagnetic?
Solution
The electronic configuration of thorium is [Rn] 5f0 6d2 7s2.
the condensed electronic configuration for its divalent cation will be
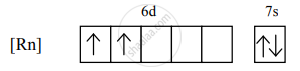
There are 2 unpaired electrons, so n = 2.
`therefore mu = sqrt(2 (2 + 2)) = sqrt8` BM = 2.83 BM
Spin only magnetic moment of the given ion is 2.83 BM. This element contains two unpaired electrons and hence, it is paramagnetic.
APPEARS IN
RELATED QUESTIONS
Answer the following
Why salts of Sc3⊕, Ti4⊕, V5⊕ are colourless?
Answer the following
Explain trends in ionisation enthalpies of d-block elements.
The catalyst used for decomposition of KClO3 is _______.
Write formula to calculate magnetic moment.
Which alloy is used in the Fischer-Tropsch process in the synthesis of gasoline?
Which of the following elements belongs to first inner transition series?
Which of the following salt contain interstitial water molecule in it?
Select the CORRECT match.
The first element of 3d series is ____________.
What is the atomicity of aluminium phosphate?
Which of the following ions will have spin-only magnetic moment equal to 1.73 BM?
Sc3+, Ti3+, Fe2+, Cu2+, Co2+
The valence shell electronic configuration of V3+ ion is ____________.
Nichrome is used specifically for making ____________.
How many among the following are ferrous alloys?
Nichrome, chromium steel, bronze, brass, stainless steel, Cupra-nickel.
Which among the following substances is used as a catalyst for thermal decomposition of potassium chlorate?
____________ has the highest electronegativity next to fluorine amongst all the elements.
What is the melting point of zinc?
Write the name of the alloy used in the Fischer Tropsch process in the synthesis of gasoline.
Salts of Ti4+ are colourless. Give reason.
Calculate spin only magnetic moment of divalent cation of transition metal with atomic number 25.
The value of the ‘spin only’ magnetic moment for one of the following configurations is 2.84 BM. The correct one is:
What is the number of unpaired electrons of nickel observed in \[\ce{[Ni(CN)4]^{2-}}\]?
The spin only magnetic moment of Cr3+ cation is ______.
