Advertisements
Advertisements
Question
Calculate the percentage of phosphorous in the fertilizer superphosphate, Ca(H2PO4)2. [Ca = 40, H =1, P =31, O = 16] (Correct to 1 decimal place)
Solution
Molecular mass of fertilizer superphosphate, Ca(H2PO4)2 =234
234 parts by weight of fertilizer contains 62 parts by weight of phosphorous
So, 100 parts will contain = 62 x 100/234 = 26.5‰
APPEARS IN
RELATED QUESTIONS
Give balanced chemical equations for the following conversions A, B, and C:
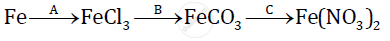
Calculate the relative molecular mass of Ammonium sulphate.
(use K = 39, Cl = 35.5, O = 16, C = 12, H = 1, Na = 23, N = 14, S= 32)
Calculate the percentage composition of oxygen in lead nitrate [Pb(NO3)2]. [Pb = 207, N= 14, O = 16]
The usefulness of a fertilizer depends upon percentage of nitrogen present in it. Find which of the following is a better fertilizer:
(a) Ammonium nitrate [NH4NO3]
(b) Ammonium phosphate [(NH4)3PO4 (N=14,H=1,O=16,P=31)
An organic compound has the following percentage composition: C = 12.76%, H = 2.13%, Br = 85.11%. The vapour density of the compound is 94. Find out its molecular formula.
Calculate the percentage of sodium in sodium aluminium fluoride (Na3AIF6).
[F = 19, Na = 23, Al = 27]
Calculate the relative molecular mass of:
Potassium chlorate
Calculate the relative molecular mass of:
(NH4)2SO4
Calculate the relative molecular mass of:
(NH4)2 Cr2O7
When heated, potassium permanganate decomposes according to the following equation :
\[\ce{2KMnO4 -> \underset{\text{solid residue}}{K2MnO4 + MnO2} + O2}\]
(a) Some potassium permanganate was heated in the test tube. After collecting one litre of oxygen at room temperature, it was found that the test tube had undergone a loss in mass of 1.32 g. If one litre of hydrogen under the same conditions of temperature and pressure has a mass of 0.0825 g, calculate the relative molecular mass of oxygen.
(b) Given that the molecular mass of potassium permanganate is 158. What volume of oxygen (measured at room temperature) would be obtained by the complete decomposition of 15.8 g of potassium permanganate? (Molar volume at room temperature is 24 litres)
