Advertisements
Advertisements
Question
Calculate spin only magnetic moment of M2+ ion.
[atomic number of M = 26]
Numerical
Solution
Atomic number (M) = 26
Electronic configuration of M atom = [Ar] 3d6 4s2
Electronic configuration of M2+ = [Ar] 3d6
So, d-orbital has the following distribution of electrons:
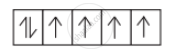
There are 4 unpaired electrons.
∴ Spin only magnetic moment (μ) = `sqrt(n(n + 2))` B.M.
= `sqrt(4(4 + 2))` B.M.
= `sqrt(4(6))` B.M.
= 4.89 B.M.
shaalaa.com
Is there an error in this question or solution?
