Advertisements
Advertisements
Question
Convert ethyne to ethane.
Solution
Ethyne is an unsaturated hydrocarbon, and when hydrogen and acetylene are passed over finely divided nickel at 200°C forms an additional compound, ethane.
\[\ce{\underset{Acetylene}{H - C ≡ C - H + H2} ->[Ni 200^\circ C] \underset{Ethene}{CH2 = CH2} ->[Ni 200^\circ C] \underset{Ethane}{CH3 - CH3}}\]
APPEARS IN
RELATED QUESTIONS
The compound formed where two alkyl groups are linked by 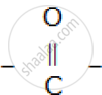 group.
group.
Write the structure of the following compound:
2 – methyl propane
What are the sources for alkynes?
What is the special feature of the structure of ethyne?
Name a triple bond hydrocarbon with two carbon atoms.
The Structure of the Following Compound :
2,3-dimethylbutane
Give the chemical equation for the preparation of ethyne from 1, 2-dibromoethane.
Name the products formed and write an equation when ethyne is added to the following in an inert solvent:
Chlorine
Give a balanced equation for the following conversion.
An alkyne to an alkene.
Substitution reactions are characteristic reactions of ______.
